Cancer associated fibroblasts in cancer development and therapy
- PMID: 40156055
- PMCID: PMC11954198
- DOI: 10.1186/s13045-025-01688-0
Cancer associated fibroblasts in cancer development and therapy
Abstract
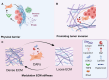
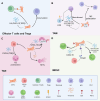
Cancer-associated fibroblasts (CAFs) are key players in cancer development and therapy, and they exhibit multifaceted roles in the tumor microenvironment (TME). From their diverse cellular origins, CAFs undergo phenotypic and functional transformation upon interacting with tumor cells and their presence can adversely influence treatment outcomes and the severity of the cancer. Emerging evidence from single-cell RNA sequencing (scRNA-seq) studies have highlighted the heterogeneity and plasticity of CAFs, with subtypes identifiable through distinct gene expression profiles and functional properties. CAFs influence cancer development through multiple mechanisms, including regulation of extracellular matrix (ECM) remodeling, direct promotion of tumor growth through provision of metabolic support, promoting epithelial-mesenchymal transition (EMT) to enhance cancer invasiveness and growth, as well as stimulating cancer stem cell properties within the tumor. Moreover, CAFs can induce an immunosuppressive TME and contribute to therapeutic resistance. In this review, we summarize the fundamental knowledge and recent advances regarding CAFs, focusing on their sophisticated roles in cancer development and potential as therapeutic targets. We discuss various strategies to target CAFs, including ECM modulation, direct elimination, interruption of CAF-TME crosstalk, and CAF normalization, as approaches to developing more effective treatments. An improved understanding of the complex interplay between CAFs and TME is crucial for developing new and effective targeted therapies for cancer.
Keywords: Cancer associated fibroblasts (CAFs); Cancer development; Cancer therapy; Extracellular matrix (ECM); Heterogeneity; Tumor microenvironment (TME).
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures




References
-
- Younesi FS, Miller AE, Barker TH, Rossi FMV, Hinz B. Fibroblast and myofibroblast activation in normal tissue repair and fibrosis. Nat Rev Mol Cell Biol. 2024;25:617–38. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

