Protocol for a single-centre randomised pilot study to assess the safety and feasibility of adding a CytoSorb filter during kidney normothermic machine perfusion to remove inflammatory and immune mediators prior to kidney transplantation
- PMID: 40157734
- PMCID: PMC11956346
- DOI: 10.1136/bmjopen-2024-093001
Protocol for a single-centre randomised pilot study to assess the safety and feasibility of adding a CytoSorb filter during kidney normothermic machine perfusion to remove inflammatory and immune mediators prior to kidney transplantation
Abstract
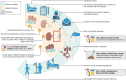
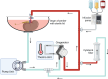
Introduction: The introduction of perfusion technologies in kidney transplantation has the potential to improve graft function and survival and increase utilisation. Our previous work demonstrated that kidneys with an enhanced inflammatory and immune response during normothermic machine perfusion (NMP) had significant graft dysfunction after transplantation. The addition of a cytokine filter (CytoSorb) to the NMP circuit dramatically reduces both circulating inflammatory mediators and inflammatory gene expression, but this has not been trialled in clinical practice.
Methods and analysis: This is a randomised phase 1 pilot study to evaluate the safety and feasibility of using a CytoSorb filter in clinical NMP to remove inflammatory and immune mediators. Eligible kidney transplant recipients on the waiting list in the East of England will be approached for consent. A total of 20 patients will be recruited and randomised in a 1:1 ratio for the donor kidney to receive either NMP or NMP with a CytoSorb filter pre-transplantation. The kidney will be transplanted according to standard practice after NMP. The primary endpoint is inflammatory and immune gene expression measured in a cortical biopsy from the kidney 60 min post-transplant. Secondary endpoints include rates and duration of delayed graft function and graft function as assessed by change in creatinine clearance and estimated glomerular filtration rate 2 days, 5 days, 1 month and 3 months post-transplant. Additionally, inflammatory mediators and injury markers will be measured in peripheral blood and urine samples taken pre-operatively and on days 2 and 5 after transplant.
Ethics and dissemination: This study has been approved by the Health Research Authority Health and Care Research Wales Committee (REC 23/WM/0141) and by National Health Service (NHS) Blood and Transplant (Ref: Study 148). Findings will be published in a peer-reviewed journal and disseminated at scientific conferences. The dataset will be made available on request.
Trial registration: The study is prospectively registered on the ISCRTN registry (ID: 13698207).
Keywords: Renal transplantation; Transplant medicine; Transplant surgery.
© Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY. Published by BMJ Group.
Conflict of interest statement
Competing interests: SH received an honorarium from CytoSorbents Europe GmbH. The other authors have no competing interests to declare. Figures used in this publication were created using Biorender.com.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
