Atp6v0d2 deficiency partially restores defects in Mcoln1-deficient mouse corpus luteum
- PMID: 40162300
- PMCID: PMC11949234
- DOI: 10.1097/RD9.0000000000000116
Atp6v0d2 deficiency partially restores defects in Mcoln1-deficient mouse corpus luteum
Abstract
Objective: ATP6V0d2 is a subunit of the vacuolar-type H+-ATPase (V-ATPase) that pumps H+ ions into lysosomes. TRPML1 (MCOLN1/Mcoln1) transports cations out of lysosomes. Mcoln1 -/- mice recapitulate the lysosomal storage disorder mucolipidosis type IV (MLIV) phenotype. We previously demonstrated that Mcoln1 -/- female mice quickly became infertile at 5 months old (5M) with degenerating corpora lutea (CL) and progesterone (P4) deficiency. We tested our hypothesis that Atp6v0d2 deficiency could partially compensate for Mcoln1 deficiency to restore CL functions in Atp6v0d2 -/- Mcoln1 -/- mice.
Methods: Control and Atp6v0d2 -/- Mcoln1 -/- female mice underwent fertility test from 2M to 7M. A subset of them was dissected at 5M on day 3.5 post-coitum (D3.5). The D3.5 ovaries from 5M control, Mcoln1 -/-, and Atp6v0d2 -/- Mcoln1 -/- mice were evaluated for CL morphology, lipid droplet staining, and markers of mitochondria and P4 steroidogenesis in the luteal cells.
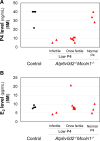
Results: The fertility test of Atp6v0d2 -/- Mcoln1 -/- female mice (2M-7M) revealed normal mating activity but reduced fertility compared with the control; yet ~25% of them remained fertile at 5M to 7M but with dystocia. We analyzed a subset of 11 Atp6v0d2 -/- Mcoln1 -/- mice (5M) in the fertility test on D3.5: three (27.3%) had normal P4 levels and all examined CL parameters, indicating full restoration of CL function compared with Mcoln1 -/-, whereas eight had P4 deficiency, with two (18.2%) infertile and six (54.5%) once fertile. In contrast to Mcoln1 -/- CLs, which had extensive amorphous cellular debris, indicating cell degeneration, Atp6v0d2 -/- Mcoln1 -/- CLs had reduced amorphous cellular debris regardless of P4 levels. However, similar to Mcoln1 -/- CLs, P4-deficient Atp6v0d2 -/- Mcoln1 -/- CLs showed impaired differentiation, enlarged lipid droplets, disorganized expression of endothelial basal lamina marker collagen IV, and reduced expression of mitochondrial marker heat shock protein 60 (HSP60) and steroidogenesis rate-limiting protein StAR, indicating that additional Atp6v0d2 deficiency compensates for Mcoln1 deficiency-induced cell degeneration, but is insufficient to restore luteal cell differentiation and P4 steroidogenesis in P4-deficient Atp6v0d2 -/- Mcoln1 -/- CLs.
Conclusion: This study shows that Atp6v0d2 -/- Mcoln1 -/- CLs had varied improvements compared with Mcoln1 -/- CLs, and it provides in vivo genetic evidence of the coordination between different lysosomal channels in CL function.
Keywords: ATP6V0D2/Atp6v0d2; Atp6v0d2−/−Mcoln1−/− mice; Corpus luteum; Progesterone; TRPML1/Mcoln1.
Copyright © 2024 Reproductive and Developmental Medicine, Published by Wolters Kluwer Health, Inc.
Conflict of interest statement
All authors declare no conflicts of interest. Xiaoqin Ye is Editorial Board member of Reproductive and Developmental Medicine. The article was subject to the journal’s standard procedures, with peer review handled independently of these Editorial Board member and their research groups.
Figures




References
-
- Mindell JA. Lysosomal acidification mechanisms. Annu Rev Physiol 2012;74:69–86. doi:10.1146/annurev-physiol-012110-142317. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
