Improvement of the diagnosis of intestinal protozoa using a multiplex qPCR strategy compared to classical microscopy: a prospective study on 3,500 stool samples over 3 years
- PMID: 40162804
- PMCID: PMC12077082
- DOI: 10.1128/jcm.01610-24
Improvement of the diagnosis of intestinal protozoa using a multiplex qPCR strategy compared to classical microscopy: a prospective study on 3,500 stool samples over 3 years
Abstract
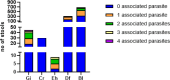
Commercial multiplex real-time PCR (qPCR assays) are now widely used for the diagnosis of intestinal protozoan infections, but few prospective studies evaluated their performances on large patient cohorts. We extracted data from our information system from 1 January 2021 to 15 March 2024 and included all stool samples analyzed in routine. Parasites were searched using a multiplex PCR (AllPlex Gastrointestinal Panel assay, Seegene) and microscopic examination with two concentration methods. Acid-fast staining was performed when Cryptosporidium detection was specifically requested. In total, 3,495 stools were analyzed from 2,127 patients. Giardia intestinalis, Cryptosporidium spp., Entamoeba histolytica, Dientamoeba fragilis, and Blastocystis spp. were found by multiplex qPCR in 45 (1.28%), 30 (0.85%), 9 (0.25%), 310 (8.86%), and 673 (19.25%) samples, respectively, alone or in combination (n = 909). In the vast majority of cases, PCR detected a protozoan on the first stool sample. Microscopy was positive for G. intestinalis, Cryptosporidium spp., Entamoeba histolytica/dispar, D. fragilis, and Blastocystis spp. in 25 (0.7%), 8 (0.23%), 24 (0.68%), 22 (0.63%), and 229 (6.55%), samples, respectively, alone or in combination (n = 286 samples). No samples were PCR-/Microscopy+ for G. intestinalis, Cryptosporidium spp., and E. histolytica, while D. fragilis and Blastocystis spp. were detected only with microscopy in 6 and 20 samples, respectively. Microscopy allowed the detection of parasites not targeted by the multiplex panel (5 Cystoisospora belli, 331 samples with non-pathogenic protozoa, and 68 samples with helminths). Overall, the multiplex PCR proved more efficient to detect protozoan parasites, but a microscopic technique should be performed when infection with C. belli (HIV-infected patients) or helminths is suspected (migrants and travelers).IMPORTANCEIn the era of increasing use of multiplex PCR panels for the diagnosis of intestinal protozoan infections, it is important to form an opinion on the positioning of those assays within a lab workflow. This study analyzes routine results obtained prospectively by microscopy and a commercial multiplex PCR over 3 years, and shows that the assay meets the expectations of a clinical laboratory for the detection of protozoan parasites of medical interest. It is recalled that Cystoisospora belli is not targeted by the multiplex assay, and that microscopy still remains necessary to detect helminths.
Keywords: diagnostics; gastrointestinal infection; intestinal parasites; molecular methods; stool.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Montresor A, Mupfasoni D, Mikhailov A, Mwinzi P, Lucianez A, Jamsheed M, Gasimov E, Warusavithana S, Yajima A, Bisoffi Z, Buonfrate D, Steinmann P, Utzinger J, Levecke B, Vlaminck J, Cools P, Vercruysse J, Cringoli G, Rinaldi L, Blouin B, Gyorkos TW. 2020. The global progress of soil-transmitted helminthiases control in 2020 and World Health Organization targets for 2030. PLoS Negl Trop Dis 14:e0008505. doi:10.1371/journal.pntd.0008505 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

