This is a preprint.
ABHD11 inhibition drives sterol metabolism to modulate T cell effector function and alleviate autoimmunity
- PMID: 40166327
- PMCID: PMC11957007
- DOI: 10.1101/2025.03.19.643996
ABHD11 inhibition drives sterol metabolism to modulate T cell effector function and alleviate autoimmunity
Abstract
Chronic inflammation in autoimmunity is driven by T cell hyperactivation. This unregulated response to self is fuelled by heightened metabolic programmes, which offers a promising new direction to uncover novel treatment strategies. α/β-hydrolase domain-containing protein 11 (ABHD11) is a mitochondrial hydrolase that maintains the catalytic function of α-ketoglutarate dehydrogenase (α-KGDH), and its expression in CD4+ T cells has been linked to remission status in rheumatoid arthritis (RA). However, the importance of ABHD11 in regulating T cell metabolism and function - and thus, the downstream implication for autoimmunity - is yet to be explored. Here, we show that pharmacological inhibition of ABHD11 dampens cytokine production by human and mouse T cells. Mechanistically, the anti-inflammatory effects of ABHD11 inhibition are attributed to increased 24,25-epoxycholesterol (24,25-EC) biosynthesis and subsequent liver X receptor (LXR) activation, which arise from a compromised TCA cycle. The impaired cytokine profile established by ABHD11 inhibition is extended to two patient cohorts of autoimmunity. Importantly, using a murine model of accelerated type 1 diabetes (T1D), we show that targeting ABHD11 suppresses cytokine production in antigen-specific T cells and delays the onset of diabetes in vivo. Collectively, our work provides pre-clinical evidence that ABHD11 is an encouraging drug target in T cell-mediated autoimmunity.
Conflict of interest statement
Competing Interests M. Niphakis is an employee of Lundbeck. All authors declare no competing interests.
Figures



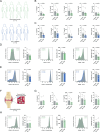

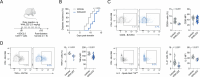
Similar articles
-
ABHD11 maintains 2-oxoglutarate metabolism by preserving functional lipoylation of the 2-oxoglutarate dehydrogenase complex.Nat Commun. 2020 Aug 13;11(1):4046. doi: 10.1038/s41467-020-17862-6. Nat Commun. 2020. PMID: 32792488 Free PMC article.
-
Human alpha beta hydrolase domain containing protein 11 and its yeast homolog are lipid hydrolases.Biochem Biophys Res Commun. 2017 Jun 10;487(4):875-880. doi: 10.1016/j.bbrc.2017.04.145. Epub 2017 Apr 29. Biochem Biophys Res Commun. 2017. PMID: 28465236
-
ABHD11 Is Critical for Embryonic Stem Cell Expansion, Differentiation and Lipid Metabolic Homeostasis.Front Cell Dev Biol. 2020 Jul 7;8:570. doi: 10.3389/fcell.2020.00570. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32733886 Free PMC article.
-
Metabolic Control of Autoimmunity and Tissue Inflammation in Rheumatoid Arthritis.Front Immunol. 2021 Apr 2;12:652771. doi: 10.3389/fimmu.2021.652771. eCollection 2021. Front Immunol. 2021. PMID: 33868292 Free PMC article. Review.
-
Optimization and characterization of a triazole urea inhibitor for alpha/beta hydrolase domain-containing protein 11 (ABHD11): anti-probe for LYPLA1/LYPLA2 dual inhibitor ML211.2011 Mar 31 [updated 2013 Mar 7]. In: Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010–. 2011 Mar 31 [updated 2013 Mar 7]. In: Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010–. PMID: 23658953 Free Books & Documents. Review.
References
-
- Gubser P.M. et al. Rapid effector function of memory CD8+ T cells requires an immediate-early glycolytic switch. Nature Immunology 14, 1064-+ (2013). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
