Research developments in the neurovascular unit and the blood‑brain barrier (Review)
- PMID: 40166412
- PMCID: PMC11956146
- DOI: 10.3892/br.2025.1966
Research developments in the neurovascular unit and the blood‑brain barrier (Review)
Abstract
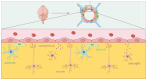
The neurovascular unit (NVU) is composed of neurons, glial cells, brain microvascular endothelial cells (BMECs), pericytes, and the extracellular matrix. The NVU controls the permeability of the blood-brain barrier (BBB) and protects the brain from harmful blood-borne and endogenous and exogenous substances. Among these, neurons transmit signals, astrocytes provide nutrients, microglia regulate inflammation, and BMECs and pericytes strengthen barrier tightness and coverage. These cells, due to their physical structure, anatomical location, or physiological function, maintain the microenvironment required for normal brain function. In this review, the BBB structure and mechanisms are examined to obtain a better understanding of the factors that influence BBB permeability. The findings may aid in safeguarding the BBB and provide potential therapeutic targets for drugs affecting the central nervous system.
Keywords: astrocyte; blood-brain barrier; brain microvascular endothelial cells; microglial; neurons; neurovascular unit; pericytes.
Copyright: © 2025 Gong et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Structure and Function of the Blood-Brain Barrier (BBB).Handb Exp Pharmacol. 2022;273:3-31. doi: 10.1007/164_2020_404. Handb Exp Pharmacol. 2022. PMID: 33249527
-
An isogenic neurovascular unit model comprised of human induced pluripotent stem cell-derived brain microvascular endothelial cells, pericytes, astrocytes, and neurons.Fluids Barriers CNS. 2019 Aug 7;16(1):25. doi: 10.1186/s12987-019-0145-6. Fluids Barriers CNS. 2019. PMID: 31387594 Free PMC article.
-
Targetability of the neurovascular unit in inflammatory diseases of the central nervous system.Immunol Rev. 2022 Oct;311(1):39-49. doi: 10.1111/imr.13121. Epub 2022 Jul 31. Immunol Rev. 2022. PMID: 35909222 Free PMC article. Review.
-
Dysfunction of the Neurovascular Unit by Psychostimulant Drugs.Int J Mol Sci. 2023 Oct 13;24(20):15154. doi: 10.3390/ijms242015154. Int J Mol Sci. 2023. PMID: 37894832 Free PMC article. Review.
-
Simple and efficient protocol to isolate and culture brain microvascular endothelial cells from newborn mice.Front Cell Neurosci. 2022 Oct 13;16:949412. doi: 10.3389/fncel.2022.949412. eCollection 2022. Front Cell Neurosci. 2022. PMID: 36313615 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
