C/EBPβ activation in vascular smooth muscle cells promotes hyperlipidemia-induced phenotypic transition and arterial stiffness
- PMID: 40169541
- PMCID: PMC11962150
- DOI: 10.1038/s41392-025-02196-w
C/EBPβ activation in vascular smooth muscle cells promotes hyperlipidemia-induced phenotypic transition and arterial stiffness
Abstract
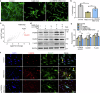
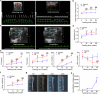
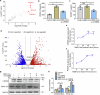
Arterial stiffness is a critical factor in cardiovascular and cerebrovascular events, yet clinical practice lacks specific therapeutic targets and biomarkers for its assessment. Hyperlipidemia closely correlates with arterial stiffness, and we observed elevated CCAAT/enhancer-binding protein β (C/EBPβ) expression in atherosclerotic mouse arterial walls. As the arterial medial layer predominantly consists of vascular smooth muscle cells (VSMCs), C/EBPβ's role in VSMCs under hyperlipidemia remains unclear. Our findings demonstrate that cholesterol-induced phenotypic transition of contractile VSMCs to macrophage-like cells coincides with C/EBPβ upregulation and activation. The activation of C/EBPβ is closely related to cellular assembly and organization, regulating the cytoskeleton via Disheveled-associated activator of morphogenesis 1 (Daam1). Conditional knockout of C/EBPβ in VSMCs of ApoE-/- mice alleviated hyperlipidemia-induced vascular remodeling and reduced the elevation of aortic pulse wave velocity. Additionally, C/EBPβ-regulated cytokine platelet-derived growth factor-CC (PDGF-CC) is correlated with brachial-ankle pulse wave velocity in humans. These results indicate that the activation of C/EBPβ promotes the transition of VSMCs from a contractile phenotype to a macrophage-like phenotype by regulating morphological changes, and C/EBPβ activation contributes to hyperlipidemia-induced arterial stiffness. PDGF-CC exhibited a significant association with arterial stiffness and may serve as a promising indicator of arterial stiffness in humans. Our study reveals molecular mechanisms behind hyperlipidemia-induced arterial stiffness and provides potential therapeutic targets and biomarkers.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures




References
-
- Barallobre-Barreiro, J. et al. Extracellular matrix in vascular disease, Part 2/4: JACC focus seminar. J. Am. Coll. Cardiol.75, 2189–2203 (2020). - PubMed
-
- Safar, M. E. Arterial stiffness as a risk factor for clinical hypertension. Nat. Rev. Cardiol.15, 97–105 (2018). - PubMed
-
- Mattace-Raso, F. U. et al. Arterial stiffness and risk of coronary heart disease and stroke: the Rotterdam Study. Circulation113, 657–663 (2006). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

