Testing an inverse modeling approach with gradient boosting regression for stroke volume estimation using patient thermodilution data
- PMID: 40171404
- PMCID: PMC11959070
- DOI: 10.3389/frai.2025.1530453
Testing an inverse modeling approach with gradient boosting regression for stroke volume estimation using patient thermodilution data
Abstract
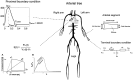
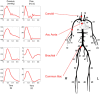
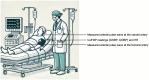
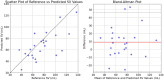
Stroke volume (SV) is a major indicator of cardiovascular function, providing essential information about heart performance and blood flow adequacy. Accurate SV measurement is particularly important for assessing patients with heart failure, managing patients undergoing major surgeries, and delivering optimal care in critical settings. Traditional methods for estimating SV, such as thermodilution, are invasive and unsuitable for routine diagnostics. Non-invasive techniques, although safer and more accessible, often lack the precision and user-friendliness needed for continuous bedside monitoring. We developed a modified method for SV estimation that combines a validated 1-D model of the systemic circulation with machine learning. Our approach replaces the traditional optimization process developed in our previous work, with a regression method, utilizing an in silico-generated dataset of various hemodynamic profiles to create a gradient boosting regression-enabled SV estimator. This dataset accurately mimics the dynamic characteristics of the 1-D model, allowing for precise SV predictions without resource-intensive parameter adjustments. We evaluated our method against SV values derived from the gold standard thermodilution method in 24 patients. The results demonstrated that our approach provides a satisfactory agreement between the predicted and reference data, with a MAE of 16 mL, a normalized RMSE of 21%, a bias of -9.2 mL, and limits of agreement (LoA) of [-47, 28] mL. A correlation coefficient of r = 0.7 (p < 0.05) was reported, with the predicted SV slightly underestimated (68 ± 23 mL) in comparison to the reference SV (77 ± 26 mL). The significant reduction in computational time of our method for SV assessment should make it suitable for real-time clinical applications.
Keywords: blood pressure; cardiac output; gradient boosting; hemodynamics; non-invasive monitoring; supervised learning.
Copyright © 2025 Bikia, Adamopoulos, Roffi, Rovas, Noble, Mach and Stergiopulos.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Derivation of Stroke Volume from Pulmonary Artery Pressures.Cardiol Ther. 2024 Jun;13(2):401-413. doi: 10.1007/s40119-024-00360-x. Epub 2024 Apr 25. Cardiol Ther. 2024. PMID: 38664318 Free PMC article.
-
Comparison of stroke volumes assessed by three-dimensional echocardiography and transpulmonary thermodilution in a pediatric animal model.J Clin Monit Comput. 2017 Apr;31(2):353-360. doi: 10.1007/s10877-016-9843-7. Epub 2016 Feb 17. J Clin Monit Comput. 2017. PMID: 26886899
-
Agreement in hemodynamic monitoring during orthotopic liver transplantation: a comparison of FloTrac/Vigileo at two monitoring sites with pulmonary artery catheter thermodilution.J Clin Monit Comput. 2017 Apr;31(2):343-351. doi: 10.1007/s10877-016-9840-x. Epub 2016 Feb 16. J Clin Monit Comput. 2017. PMID: 26884378
-
Noninvasive cardiac output measurement in heart failure subjects on circulatory support.Anesth Analg. 2009 Mar;108(3):881-6. doi: 10.1213/ane.0b013e318193174b. Anesth Analg. 2009. PMID: 19224797 Review.
-
New Developments in Hemodynamic Monitoring.J Cardiothorac Vasc Anesth. 2019 Aug;33 Suppl 1:S67-S72. doi: 10.1053/j.jvca.2019.03.043. J Cardiothorac Vasc Anesth. 2019. PMID: 31279355 Review.
References
-
- Bikia V., Pagoulatou S., Trachet B., Soulis D., Protogerou A. D., Papaioannou T. G., et al. . (2020). Noninvasive cardiac output and central systolic pressure from cuff-pressure and pulse wave velocity. IEEE J. Biomed. Health Inform. 24, 1968–1981. doi: 10.1109/JBHI.2019.2956604, PMID: - DOI - PubMed
-
- Bikia V., Rovas G., Pagoulatou S., Stergiopulos N. (2021). Determination of aortic characteristic impedance and total arterial compliance from regional pulse wave velocities using machine learning: an in-silico study. Front. Bioeng. Biotechnol. 9:649866. doi: 10.3389/fbioe.2021.649866, PMID: - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

