Evaluation of phenotypic and genotypic methods for detecting KPC variants
- PMID: 40178306
- PMCID: PMC12057356
- DOI: 10.1128/aac.00082-25
Evaluation of phenotypic and genotypic methods for detecting KPC variants
Abstract
Klebsiella pneumoniae carbapenemases (KPCs) have spread and diversified extensively. To date, 242 clinical variants have been identified and harbor different hydrolytic capacities, thereby interfering with rapid diagnostic tests. The accurate detection of KPC variants is crucial to guide treatment and control measures in healthcare settings. We constructed KPC variants to assess the mutational impact on detection capacities of resistance-based tests. KPC variants (n = 45) were characterized phenotypically and used to measure the detection sensitivity of KPC detection methods (two lateral flow immunoassays [LFIAs], three hydrolysis tests, three selective culture media, and two PCR-based tests). We identified four antibiotic susceptibility patterns: "KPC-like" (23/45; 51%), "extended-spectrum beta-lactamase-like" (6/45; 13%), "ceftazidimase" (9/45; 20%), and outlier variants with "mixed-profiles" (5/45; 11%). These phenotypes had different impacts on the detection capabilities of hydrolysis tests (0%-100%), LFIA (44%-100%), and selective culture media (0%-100%), highlighting a risk of misdiagnosis for some KPC variants. All variants were detected with PCR-based tests. To detect the maximum of KPC variants, fecal carriage screening requires a combination of selective media targeting resistance to carbapenems, third-generation cephalosporins, and ceftazidime-avibactam. From antibiotic susceptibility testing, resistance to ceftazidime ± avibactam and specific phenotypic profiles should be used as warnings to track the presence of KPC variants. We recommend LFIA as a first-line test, owing to its high sensitivity in detecting KPC variants. Nevertheless, using a combination of tests may remain wise in some situations. The spread of KPC variants remains a significant concern, particularly as reversion to ancestral phenotype could restore carbapenem resistance and lead to therapeutic failure.
Keywords: KPC beta-lactamase; LFIA tests; clinical KPC variants; detection tests; hydrolysis-based tests.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
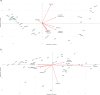
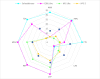
References
-
- Zhanel GG, Lawson CD, Adam H, Schweizer F, Zelenitsky S, Lagacé-Wiens PRS, Denisuik A, Rubinstein E, Gin AS, Hoban DJ, Lynch JP, Karlowsky JA. 2013. Ceftazidime-avibactam: a novel cephalosporin/β-lactamase inhibitor combination. Drugs (Abingdon Engl) 73:159–177. doi:10.1007/s40265-013-0013-7 - DOI - PubMed
-
- Zhanel GG, Lawrence CK, Adam H, Schweizer F, Zelenitsky S, Zhanel M, Lagacé-Wiens PRS, Walkty A, Denisuik A, Golden A, Gin AS, Hoban DJ, Lynch JP, Karlowsky JA. 2018. Imipenem–relebactam and meropenem–vaborbactam: two novel carbapenem-β-lactamase inhibitor combinations. Drugs (Abingdon Engl) 78:65–98. doi:10.1007/s40265-017-0851-9 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

