Design, development, and preclinical evaluation of pirfenidone-loaded nanostructured lipid carriers for pulmonary delivery
- PMID: 40181013
- PMCID: PMC11968824
- DOI: 10.1038/s41598-025-90910-7
Design, development, and preclinical evaluation of pirfenidone-loaded nanostructured lipid carriers for pulmonary delivery
Abstract
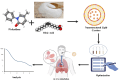
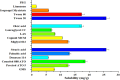
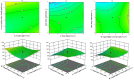
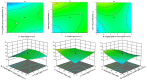
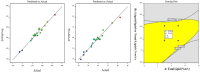
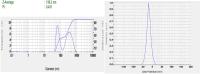
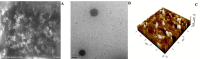
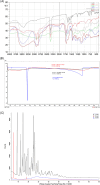
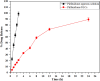
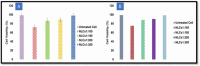
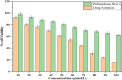
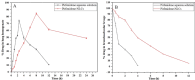
Pirfenidone is an antifibrotic and anti-inflammatory drug used for the management of idiopathic pulmonary fibrosis. The current oral delivery of PD has multiple drawbacks, including first-pass metabolism and gastrointestinal discomfort. Efforts have been made to create nanostructured lipid carriers (NLCs) using solid lipids, liquid lipids, and surfactants through an emulsification process followed by ultrasonication to achieve sustained drug release. A central composite design (CCD) utilizing response surface methods (RSMs) was employed to develop and optimize the formulation. The assessed characteristics included particle size distribution, surface topography, drug entrapment efficiency, in vitro drug release, and kinetic profiles in animal models. Cytotoxicity experiments were performed on HepG2 and Caco-2 cell lines and compared with that of PD-NLCs. The optimized formulation yielded a particle size of 159.8 ± 3.46 nm and an encapsulation efficiency of 81.4 ± 7.1% after 10 freeze-thaw cycles of homogenized lipid carriers. In vitro tests assessing various tested flow rates revealed that over 95% of the released drug was retrieved. In vitro studies showed that the PD-loaded nanostructured lipid carrier (NLC) was more cytotoxic to HepG2 and Caco-2 cells than a pure aqueous solution of the drug. Using 25% w/w sorbitol as a cryoprotectant, the findings showed no variation in the properties of NLC before and after freeze-drying. PD-NLCs carriers were shown to have better bioavailability, longer retention time in the lung, and a 15.94-targeting factor related to the PD aqueous solution. Hence, the outcomes confirmed the potential of the PD-NLCs formulation to improve the efficacy of the drug in inhalation therapy.
Keywords: Bioavailability; Caco2; Central composite design; HepG2; Lung targeting; Nanostructured lipid carriers; Pirfenidone; Pulmokinetic parameters.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The Institutional Animal Ethics Committee (IAEC) of Andhra University in Visakhapatnam (IAEC No: I/IAEC/AU/013/2021WR) approved our testing technique, which was developed under the CPCSEA guidelines. The in vivo procedure was performed according to the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA). All in vivo studies complied with the ARRIVE criteria. This study was conducted in accordance with ARRIVE guidelines. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures
References
-
- Hilberg, O., Simonsen, U., du Bois, R. & Bendstrup, E. Pirfenidone: Significant treatment effects in idiopathic pulmonary fibrosis. Clin. Respir. J.6, 131–143 (2012). - PubMed
-
- Richeldi, L., Yasothan, U., Kirkpatrick, P. & Pirfenidone Nat. Rev. Drug Discov.10, 489–491 (2011). - PubMed
-
- Taniguchi, H. et al. Pirfenidone in idiopathic pulmonary fibrosis. Eur. Respir. J.35, 821–829 (2010). - PubMed
-
- Carter, N. J. Pirfenidone Drugs71, 1721–1732 (2011). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

