Does adjuvant chemotherapy result in poorer health-related quality of life among colorectal cancer patients? A longitudinal multisite observational study in Singapore
- PMID: 40186288
- PMCID: PMC11971881
- DOI: 10.1186/s12955-025-02363-1
Does adjuvant chemotherapy result in poorer health-related quality of life among colorectal cancer patients? A longitudinal multisite observational study in Singapore
Abstract
Background: Research on health-related quality of life (HRQOL) using minimally important differences for cancer care in Asian settings are sparse. This study aimed to describe functional HRQOL trajectories among Colorectal Cancer (CRC) patients undergoing adjuvant chemotherapy (AC) compared to those who did not (No AC), evaluate if AC was associated with change in HRQOL prospectively, and examine QOL differences between elderly and non-elderly CRC patients requiring AC.
Methods: CRC patients diagnosed between February 2018 to August 2021 were recruited from three Singapore public hospitals. Participants completed the EORTC QLQ-C30 over seven timepoints (diagnosis, predischarge, 1-, 3-, 6-, 9-, 12-months post-surgery). Clinical characteristics were collected from electronic medical records.
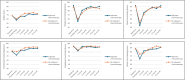
Results: The sample comprised 251 participants (102 in AC group; 40.64%). Clinically relevant deteriorations in functional HRQOL were observed in both groups between baseline and predischarge. These returned to baseline by 12-month. AC was associated with poorer physical (β = -35.34, p < 0.05) and role functioning (β = -71.17, p < 0.05) over time. Being elderly was associated with poorer physical functioning (β = -0.44, p < 0.05) over time. However, the non-elderly AC subgroup tended to experience poorer HRQOL in general compared to elderly.
Conclusions: Functional recovery remains a challenge for CRC patients in general. However, non-elderly AC patients may experience more severe impacts to role and social functioning.
Keywords: Adjuvant; Chemotherapy; Colorectal neoplasms; Quality of life.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Approval to proceed with this study was provided by the National Healthcare Group’s Domain Specific Review Board (NHG DSRB Ref: 2017/00518) in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients participating in this study. Consent for publication: N/A. Competing interests: The authors declare no competing interests.
Figures
References
-
- Baidoun F, Elshiwy K, Elkeraie Y, Merjaneh Z, Khoudari G, Sarmini MT, et al. Colorectal cancer epidemiology: recent trends and impact on outcomes. Curr Drug Targets. 2021;22(9):998–1009. - PubMed
-
- National Cancer Institute. Colon cancer treatment (PDQ®)–health professional version. 2025. Available from: https://www.cancer.gov/types/colorectal/hp/colon-treatment-pdq#_45.
-
- National Cancer Institute. Rectal cancer treatment (PDQ®)–health professional version. 2025. Available from: https://www.cancer.gov/types/colorectal/hp/rectal-treatment-pdq#_43.
-
- American Cancer Society. Chemotherapy for colorectal cancer. 2024. Available from: https://www.cancer.org/cancer/types/colon-rectal-cancer/treating/chemoth....
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical