Burden of eosinophilic esophagitis in adult and adolescent patients: results from a real-world analysis
- PMID: 40188493
- PMCID: PMC11972634
- DOI: 10.1093/dote/doaf024
Burden of eosinophilic esophagitis in adult and adolescent patients: results from a real-world analysis
Abstract
Background: Real-world data on the impact of eosinophilic esophagitis (EoE) in patients are limited. This study assessed clinical characteristics, healthcare resource utilization (HCRU), symptoms, comorbidities, and quality of life (QoL) of EoE patients.
Methods: The multicountry cross-sectional survey, Adelphi EoE Disease Specific Programme™, collected physician and patient-reported data on clinical characteristics, HCRU, symptoms, comorbidities, and QOL of EoE patients with past/current proton pump inhibitor use and ongoing dysphagia-related symptoms (September to December 2020) at study entry.
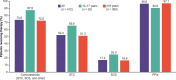
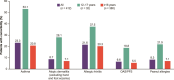
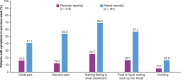
Results: Physicians provided clinical characteristics, symptom, comorbidity, and HCRU data for 412 patients (12-17 years: 8%; ≥18 years: 92%); and 161 of these patients (12-17 years: 6%; ≥18 years: 94%) provided symptom and QOL data. Of the 412 patients, 67% were male, with a mean (SD) age of 37.0 (15.3) years. Overall, 74% of patients were currently being treated with corticosteroids (12-17 years: 88%; ≥18 years: 73%); 25% of patients had a history of esophageal dilations (12-17 years: 19%; ≥18 years: 26%); and 30% of patients had EoE-related emergency room visit (12-17 years: 31%; ≥18 years: 30%) in the last year. Among the 161 patients, heartburn (69%) was the most commonly reported symptom; the greatest negative impacts on QOL were reported for dysphagia-related anxiety, social activities involving food, and maintaining friendships (EoE Impact Questionnaire scores [1-5, low to high impact]: 1.6-2.2 for both age groups).
Conclusion: EoE patients continued to experience disease burden despite receiving treatment, highlighting the high unmet need for effective disease management in this population.
Keywords: clinical burden; eosinophilic esophagitis; healthcare resource utilization; quality of life; real-world evidence.
© The Author(s) 2025. Published by Oxford University Press on behalf of International Society for Diseases of the Esophagus.
Figures

References
-
- Mukkada V, Falk G W, Eichinger C S, King D, Todorova L, Shaheen N J. Health-related quality of life and costs associated with eosinophilic esophagitis: a systematic review. Clin Gastroenterol Hepatol 2018; 16: 495–503.e8. - PubMed
-
- Schoepfer A M, Safroneeva E, Bussmann C et al. Delay in diagnosis of eosinophilic esophagitis increases risk for stricture formation in a time-dependent manner. Gastroenterology 2013; 145: 1230–6.e1-2. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

