Hand hygiene versus additional non-sterile gloves and gowns use to prevent sepsis in preterm infants colonized with multi-resistant drug bacteria: the study protocol of the cluster-randomized, cross-over, non-inferiority BALTIC trial
- PMID: 40189707
- PMCID: PMC11973001
- DOI: 10.1186/s40348-025-00192-2
Hand hygiene versus additional non-sterile gloves and gowns use to prevent sepsis in preterm infants colonized with multi-resistant drug bacteria: the study protocol of the cluster-randomized, cross-over, non-inferiority BALTIC trial
Abstract
Background: Infections are highly relevant for neonatal mortality and long-term morbidities in survivors. Therefore, it is an urgent need to optimize and evaluate infection prevention and control (IPC) strategies. Several infection outbreaks in German neonatal intensive care units (NICUs) required rapid responses by hospitals and improved future preparedness. As a consequence, German authorities recommended weekly colonization screening on NICUs. This screening aims to detect multidrug-resistant organisms (MDRO) and bacteria with high transmissibility. According to these guidelines, infants colonized with multiresistant gram-negative (MRGN) bacteria with in-vitro resistance to piperacillin and cephalosporins (2MRGN) should be cared wearing non-sterile gloves and gowns in addition to standard hygiene precautions. Whether these extended IPC measures have an individual benefit for infants or contribute to the prevention of infection outbreaks has not yet been scientifically proven. This study aims to evaluate the effect of hand desinfection as compared to hand desinfection + gloves and gowns (barrier care) for the care of 2MRGN colonized infants in NICUs on infection and transmission rates through a multicenter, cluster randomized controlled trial (BALTIC study, Barrier protection to lower transmission and infection rates with Gram-negative 2-MRGN in preterm children).
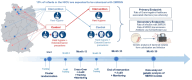
Methods: 12 participating NICUs were randomly allocated to two trial arms: receiving the intervention "standard precautions with a special focus on hand desinfection" or control (standard precautions "plus" barrier care) for the care of 2MRGN positive infants. Cross over was performed after 12 months for another 12 months per site. Primary outcome was the rate of healthcare-associated (HA) Gram-negative bloodstream infections. Secondary outcomes included transmission rate with screening relevant bacteria, overall rate of clinical and culture-proven infections, number of antibiotic cycles and desinfectant use. Regular trainings and hygiene audits are standardized co-interventions.
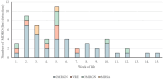
Benchmarking results: According to our single center data, 9.3% of NICU-treated infants are colonized with 2MRGN during their hospital stay. BALTIC randomized the first center in October 2020 and finished data collection including close-out monitoring in January 2024. Data analysis will be completed in May 2025.
Conclusions: BALTIC should contribute to better evidence on the effectiveness of hand desinfection and extended barrier precautions in critically ill newborns. Further benefits include comprehensive multi-center data collection on MDRO colonization dynamics, an improved awareness on IPC strategies and establishment of network platforms including antimicrobial stewardship programs.
Keywords: Gown; Hand hygiene; Multi-drug-resistant organisms; Non-sterile glove use; Preterm infants; Sepsis.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The BALTIC study was approved by the institutional review board of the University of Lübeck (primary vote, 19–275) and by the review boards of all participating sites (secondary vote). No identifying data about the infants were collected. Consent for publication: Not applicable. Competing interests: JP received a grant from the German ministry of education and research (#01EO2106). MK has received honoraria for consulting, presentations, expert testimony, and advisory board participation from Abbott, GSK, Pfizer, and Sanofi outside the submitted work. He is member in the board of the AWMF-guidelines “sustainability in intensive care and emergency medicine” and “invasive ventilation and use of extracorporeal oxygenation in patients with respiratory failure” outside the submitted work and has received travel support from the Deutsche Gesellschaft für Hygiene und Mikrobiologie.The other authors have no conflicts of interest relevant to this article to disclose. Trial registration number: DRKS00019103.
Figures
References
-
- Humberg A, Härtel C, Rausch TK, Stichtenoth G, Jung P, Wieg C, Kribs A, von der Wense A, Weller U, Höhn T, Olbertz DM, Felderhoff-Müser U, Rossi R, Teig N, Heitmann F, Schmidtke S, Bohnhorst B, Vochem M, Segerer H, Möller J, Eichhorn JG, Wintgens J, Böttger R, Hubert M, Dördelmann M, Hillebrand G, Roll C, Jensen R, Zemlin M, Mögel M, Werner C, Schäfer S, Schaible T, Franz A, Heldmann M, Ehlers S, Kannt O, Orlikowsky T, Gerleve H, Schneider K, Haase R, Böckenholt K, Linnemann K, Herting E, Göpel W (2020) Active perinatal care of preterm infants in the German Neonatal Network. Archives of disease in childhood. Fetal and neonatal edition 105, 190–195 - PubMed
-
- Härtel C, Faust K, Fortmann I, Humberg A, Pagel J, Haug C, Kühl R, Bohnhorst B, Pirr S, Viemann D, Simon A, Zemlin M, Poralla S, Müller A, Köstlin-Gille N, Gille C, Heckmann M, Rupp J, Herting E, Göpel W (2020) Sepsis related mortality of extremely low gestational age newborns after the introduction of colonization screening for multi-drug resistant organisms. Antimicrobial resistance and infection control 9, 144 - PMC - PubMed
-
- Mukhopadhyay S, Puopolo KM, Hansen NI, Lorch SA, DeMauro SB, Greenberg RG, Cotten CM, Sanchez PJ, Bell EF, Eichenwald EC, Stoll BJ (2021) Neurodevelopmental outcomes following neonatal late-onset sepsis and blood culture-negative conditions. Archives of disease in childhood. Fetal and neonatal edition 106, 467–473 - PMC - PubMed
-
- Schwartz DJ, Shalon N, Wardenburg K, DeVeaux A, Wallace MA, Hall-Moore C, Ndao IM, Sullivan JE, Radmacher P, Escobedo M, Burnham CD, Warner BB, Tarr PI, Dantas G (2023) Gut pathogen colonization precedes bloodstream infection in the neonatal intensive care unit. Sci Transl Med 15:eadg5562 - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

