SAP30, a novel autophagy regulatory gene in neuroblastoma
- PMID: 40190355
- PMCID: PMC11969447
- DOI: 10.1016/j.omton.2024.200916
SAP30, a novel autophagy regulatory gene in neuroblastoma
Abstract
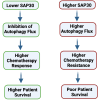
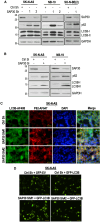
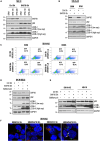
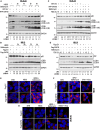
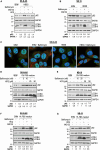
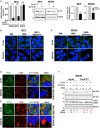
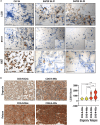
Neuroblastoma (NB), a devastating pediatric cancer originating from neural crest cells crucial for nervous system development, poses a significant therapeutic challenge. Despite chemotherapy being the primary treatment, approximately 70% of high-risk NB cases develop resistance. Autophagy is vital for neuronal development, balance, and differentiation of neural stem cells into mature neurons. However, the intricate mechanisms governing autophagy and the pivotal genes orchestrating its regulation in NB remain largely elusive. In this study, we first identified Sin3A Associated Protein 30 (SAP30) as a novel regulator of autophagy in NB. Silencing SAP30 inhibits autophagy and disrupts starvation-induced physiological autophagy in NB cells. Conversely, ectopic expression of SAP30 induces autophagy in NB cells under normal or starvation conditions. Mechanistically, SAP30 transcriptionally regulates STX17, a crucial protein involved in autophagosome-lysosome fusion during autophagy. Reduction of SAP30 decreases STX17 expression, hindering its translocation to the autophagic membrane and inhibiting autophagosome-lysosome fusion. SAP30-mediated autophagy enhances cell growth and provides protection in NB cells treated with chemotherapy drugs. Notably, suppressing SAP30 in vivo increases LC3B accumulation, an autophagy marker, along with reduced proliferation markers, both in vivo and in PDX tumors. Therefore, SAP30 emerges as a potential target to enhance NB responsiveness to chemotherapy drugs.
Keywords: MT: Novel therapeutic targets and biomarker development special issue; SAP30; autophagosome; autophagy; chemotherapy response; neuroblastoma.
© 2024 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
LinkOut - more resources
Full Text Sources
