Activating Striatal Parvalbumin Interneurons to Alleviate Chemotherapy-Induced Muscle Atrophy
- PMID: 40196908
- PMCID: PMC11976163
- DOI: 10.1002/jcsm.13782
Activating Striatal Parvalbumin Interneurons to Alleviate Chemotherapy-Induced Muscle Atrophy
Abstract
Background: Cisplatin is a widely used chemotherapeutic agent for treating solid tumours. Still, it induces severe side effects, including muscle atrophy. Understanding the mechanisms of cisplatin-induced muscle loss and exploring potential therapeutic strategies are essential. Parvalbumin (PV) interneurons in the striatum play a crucial role in motor control, and recent studies suggest that their activation may alleviate motor deficits. This study investigates the effects of chemogenetic activation of PV interneurons on cisplatin-induced muscle atrophy and motor dysfunction in mice.
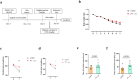
Methods: Wild-type C57BL/6 mice and transgenic hM3Dq mice were used in this study. Cisplatin (3 mg/kg) was administered intraperitoneally for 7 days to induce muscle atrophy. Mice were then treated with clozapine-n-oxide (CNO) to activate PV interneurons. Muscle strength and endurance were assessed using grip strength measurements, the inverted grid test and the wire hang test. Neuromuscular junction (NMJ) integrity was examined via histological analysis. Exercise intervention was also included, using a treadmill with a 15° incline for 60 min at varying speeds during seven consecutively days.
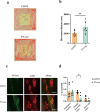
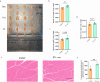
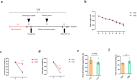
Results: Cisplatin treatment significantly reduced body weight (p < 0.001), grip strength (forelimb strength: p < 0.001, four-limb strength: p < 0.001), endurance (inverted grid test: p = 0.047, wire hang test: p = 0.014) and NMJ integrity (partially innervated NMJs: p = 0.0383). PV interneuron activation with CNO improved spontaneous motor activity in cisplatin-treated mice, as evidenced by a significant increase in total travel distance (p = 0.049) in the open-field test. Histological analysis showed a reduced ratio of partially innervated NMJs in the PV-cre group compared to the control virus group (p = 0.0441). Muscle strength also improved significantly, with forelimb grip strength increased (p < 0.001) and four-limb grip strength increased (p = 0.018). Muscle wet-weight ratios were significantly higher in the PV-cre group (quadriceps: p = 0.030). Exercise intervention significantly improved grip strength (forelimb: p < 0.001, four-limb: p = 0.002), muscle endurance (four-limb hang test: p = 0.048) and muscle weight (quadriceps: p = 0.015, gastrocnemius: p = 0.022), with an increase in muscle fibre cross-sectional area (p = 0.0018).
Conclusion: Activation of PV interneurons significantly alleviates cisplatin-induced motor deficits and muscle atrophy by improving spontaneous motor activity, NMJ integrity and muscle function. It has a similar effect to short-term exercise and may offer a promising therapeutic strategy for mitigating chemotherapy-induced muscle atrophy.
Keywords: cisplatin; muscle atrophy; parvalbumin interneurons; striatum.
© 2025 The Author(s). Journal of Cachexia, Sarcopenia and Muscle published by Wiley Periodicals LLC.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Obesity-induced type 2 diabetes impairs neurological recovery after stroke in correlation with decreased neurogenesis and persistent atrophy of parvalbumin-positive interneurons.Clin Sci (Lond). 2019 Jul 1;133(13):1367-1386. doi: 10.1042/CS20190180. Print 2019 Jul 15. Clin Sci (Lond). 2019. PMID: 31235555
-
Capsaicin alleviates cisplatin-induced muscle loss and atrophy in vitro and in vivo.J Cachexia Sarcopenia Muscle. 2023 Feb;14(1):182-197. doi: 10.1002/jcsm.13120. Epub 2022 Nov 18. J Cachexia Sarcopenia Muscle. 2023. PMID: 36401337 Free PMC article.
-
Deletion of Transferrin Receptor 1 in Parvalbumin Interneurons Induces a Hereditary Spastic Paraplegia-Like Phenotype.J Neurosci. 2023 Jul 5;43(27):5092-5113. doi: 10.1523/JNEUROSCI.2277-22.2023. Epub 2023 Jun 12. J Neurosci. 2023. PMID: 37308296 Free PMC article.
-
Chronic nicotine impairs sparse motor learning via striatal fast-spiking parvalbumin interneurons.Addict Biol. 2021 May;26(3):e12956. doi: 10.1111/adb.12956. Epub 2020 Aug 6. Addict Biol. 2021. PMID: 32767546 Free PMC article.
-
Cisplatin-Induced Muscle Wasting and Atrophy: Molecular Mechanism and Potential Therapeutic Interventions.J Cachexia Sarcopenia Muscle. 2025 Jun;16(3):e13817. doi: 10.1002/jcsm.13817. J Cachexia Sarcopenia Muscle. 2025. PMID: 40343378 Free PMC article. Review.
References
-
- Blauwhoff‐Buskermolen S., Versteeg K. S., de van der Schueren M. A., et al., “Loss of Muscle Mass During Chemotherapy Is Predictive for Poor Survival of Patients With Metastatic Colorectal Cancer,” Journal of Clinical Oncology 34, no. 12 (2016): 1339–1344. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

