Long-term metabolic changes with bictegravir/emtricitabine/tenofovir alafenamide or dolutegravir-containing regimens for HIV
- PMID: 40197415
- PMCID: PMC11978100
- DOI: 10.1186/s12981-025-00732-w
Long-term metabolic changes with bictegravir/emtricitabine/tenofovir alafenamide or dolutegravir-containing regimens for HIV
Abstract
Background: To evaluate long-term changes in weight and metabolic parameters in people with HIV-1 (PWH) initiating first-line antiretroviral therapy.
Methods: Analysis of two Phase 3, randomized, double-blind, active-controlled trials (1489: NCT02607930; 1490: NCT02607956). PWH received bictegravir/emtricitabine/tenofovir alafenamide (B/F/TAF) or dolutegravir (DTG)-based treatment (Study 1489: dolutegravir/abacavir/lamivudine [DTG/ABC/3TC]; Study 1490: DTG + F/TAF) for 144 weeks, followed by B/F/TAF (96-week open-label extension up to Week 240). Weight and metabolic parameters were assessed through Week 144 by randomized treatment assignment. Weight changes by baseline viral load and CD4 count were evaluated in PWH receiving B/F/TAF from baseline through Week 240. Multivariate modeling explored baseline factors associated with absolute weight and weight change through Week 240 and weight gain ≥ 10% at Week 240.
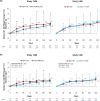
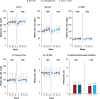
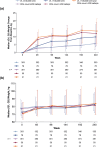
Results: Median weight and body mass index (BMI) increased over time with B/F/TAF (n = 628), DTG/ABC/3TC (n = 315), and DTG + F/TAF (n = 325). There were no significant differences in change in weight or BMI between the B/F/TAF and DTG + F/TAF groups or between the B/F/TAF and DTG/ABC/3TC groups at Week 144 in either trial, nor were there differences in other metabolic parameters, including incidence of treatment-emergent diabetes mellitus and hypertension through Week 144. Among PWH receiving B/F/TAF (baseline through Week 240), weight increases were greatest soon after initiating antiretroviral therapy (i.e., Weeks 0-48), particularly in participants with baseline viral load > 100,000 copies/ml and/or CD4 count < 200 cells/µl. In multivariate modeling (B/F/TAF pooled data), lower baseline CD4 count and higher HIV-1 RNA were associated with lower baseline weight and greater weight gain, but not absolute weight, from Week 48 through Week 240.
Conclusions: No significant difference in weight change from baseline to Week 144 was found between bictegravir and DTG, or between B/F/TAF and a non-TAF-containing regimen, in these two randomized trials. Furthermore, weight gain following treatment initiation was greatest in the first year of treatment and most pronounced in individuals with more advanced HIV at baseline, supporting the hypothesis that weight gain following initial treatment is linked to a "return to health" in people with advanced HIV.
Keywords: Antiretroviral therapy; Bictegravir; Body mass index; Dolutegravir; Glucose; HIV-1; Tenofovir alafenamide.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Both studies were undertaken in accordance with the Declaration of Helsinki and were approved by central or site-specific review boards or ethics committees. All participants provided written informed consent. Consent for publication: Not applicable. Prior presentation: Data were partially previously presented as an oral presentation at IDWeek 2021 (virtual), September 29–October 3, and in poster format at IDWeek 2022, October 19–23, Washington, DC, USA. Competing interests: E.S.D. has received research support from Gilead Sciences, Inc. and ViiV Healthcare; and consultancy fees from Gilead Sciences, Inc., Genentech, and ViiV Healthcare. C.O. has received grants or contracts from ViiV Healthcare (paid to institution); speaker honoraria from Gilead Sciences, Inc., GSK, MSD, and ViiV Healthcare; and support for attending meetings and/or travel from Gilead Sciences, Inc.; and is a governing council member of the International AIDS Society (unpaid). P.E.S. has received research support from Gilead Sciences, Inc., and ViiV Healthcare; and consultancy fees from AbbVie, Gilead Sciences, Inc., Merck, and ViiV Healthcare. D.H. has no conflicts of interest to disclose. A.P. has received grants or contracts from Gilead Sciences, Inc., and ViiV Healthcare (paid to institution); payment or honoraria for lectures, presentations, or educational events from Gilead Sciences, Inc., and ViiV Healthcare; and support for attending meetings and/or travel from Gilead Sciences, Inc., and ViiV Healthcare; and is President of NEAT ID and a member of guideline committees for BHIVA and EACS. K.W. has received research support from Gilead Sciences, Inc. C.B. has received speaker honoraria from Gilead Sciences, Inc.; and support for attending meetings and/or travel from Gilead Sciences, Inc. J.M.T. has received grants or contracts from MSD; consulting fees and speaker honoraria from Gilead Sciences, Inc., Janssen, MSD, and ViiV Healthcare; and support for attending meetings and/or travel from Gilead Sciences, Inc., Janssen, and MSD; and has participated on a data safety monitoring board or advisory board for Gilead Sciences, Inc., Janssen, MSD, and ViiV Healthcare. H.L., C.D., C.C., S.M., and J.T.H. are employees of, and hold stock/stock options in, Gilead Sciences, Inc. M.R. has received consulting fees from AbbVie, Gilead Sciences, Inc., Merck, and ViiV Healthcare; and speaker honoraria from AbbVie, Gilead Sciences, Inc., and ViiV Healthcare.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

