Brain Volume Loss After Stereotactic Laser Interstitial Thermal Therapy in Patients With Temporal Lobe Epilepsy
- PMID: 40197718
- PMCID: PMC11977048
- DOI: 10.1111/jon.70039
Brain Volume Loss After Stereotactic Laser Interstitial Thermal Therapy in Patients With Temporal Lobe Epilepsy
Abstract
Background and purpose: Temporal lobe epilepsy is the most common form of focal epilepsy. MR-guided laser interstitial thermal therapy (LITT) of the amygdalohippocampal complex has become an established therapy option in case of drug resistance. Long-term anatomic network effects on the brain due to deafferentiation have not yet been evaluated.
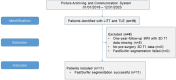
Methods: We analyzed brain volumes of 11 patients with temporal lobe epilepsy before and 1-year after hippocampal LITT with FastSurfer segmenting T1-weighted data. Additionally, we performed visual ratings and measurements.
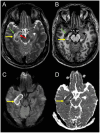
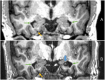
Results: A total of 11 patients with temporal lobe epilepsy (7 left-sided, 4 right-sided) were included (5 females); the mean age years (±standard deviation) at surgery was 41.5 (±18.4) years. The mean postoperative defect size was 1427 (±517) mm3. Volumetry as well as visual ratings found a progressive volume loss after left-sided surgery in the ipsilateral temporal lobe, the contralateral (right) part of the thalamus, and especially contralateral (right) fusiform cortex. These changes could not be detected for right-sided surgery.
Conclusion: A (partial) ablation of the left (dominant) hippocampus appears to exert long-term effects on the right thalamus and right-sided temporal cortices. However, we could not observe this effect in the reverse direction. Volumetric studies for larger cohorts should be conducted to investigate these findings.
Keywords: FastSurfer; epilepsy; hippocampal region; laser interstitial thermal therapy; volumetry.
© 2025 The Author(s). Journal of Neuroimaging published by Wiley Periodicals LLC on behalf of American Society of Neuroimaging.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Laser interstitial thermal therapy for medically intractable mesial temporal lobe epilepsy.Epilepsia. 2016 Feb;57(2):325-34. doi: 10.1111/epi.13284. Epub 2015 Dec 24. Epilepsia. 2016. PMID: 26697969
-
Improved operative efficiency using a real-time MRI-guided stereotactic platform for laser amygdalohippocampotomy.J Neurosurg. 2018 Apr;128(4):1165-1172. doi: 10.3171/2017.1.JNS162046. Epub 2017 Jun 30. J Neurosurg. 2018. PMID: 28665249
-
Ablation of apparent diffusion coefficient hyperintensity clusters in mesial temporal lobe epilepsy improves seizure outcomes after laser interstitial thermal therapy.Epilepsia. 2023 Mar;64(3):654-666. doi: 10.1111/epi.17432. Epub 2023 Jan 12. Epilepsia. 2023. PMID: 36196769
-
A neurosurgeon`s view: Laser interstitial thermal therapy of mesial temporal lobe structures.Epilepsy Res. 2018 May;142:135-139. doi: 10.1016/j.eplepsyres.2017.10.015. Epub 2017 Oct 27. Epilepsy Res. 2018. PMID: 29111296 Review.
-
Laser Interstitial Thermal Therapy for Mesial Temporal Lobe Epilepsy.Neurosurgery. 2016 Dec;79 Suppl 1:S83-S91. doi: 10.1227/NEU.0000000000001439. Neurosurgery. 2016. PMID: 27861328 Review.
References
-
- Slinger G., Otte W. M., Braun K. P. J., and Van Diessen E., “An Updated Systematic Review and Meta‐Analysis of Brain Network Organization in Focal Epilepsy: Looking Back and Forth,” Neuroscience and Biobehavioral Reviews 132 (2022): 211–223. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

