The dynamics of chemoattractant receptors redistribution in the electrotaxis of 3T3 fibroblasts
- PMID: 40200280
- PMCID: PMC11980103
- DOI: 10.1186/s12964-025-02165-4
The dynamics of chemoattractant receptors redistribution in the electrotaxis of 3T3 fibroblasts
Abstract
Background: Electrotaxis, the directed cell movement in direct current electric field (dcEF), is crucial for wound healing and development. We recently proposed a biphasic electrotaxis mechanism, where an initial rapid response is driven by ionic mechanisms, while redistribution of membrane components come into play during prolonged exposure to dcEF.
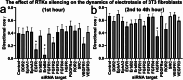
Methods: To verify this hypothesis, we studied the redistribution dynamics of EGFR, PDGFRα/β, and TGFβR1 in dcEF. For this purpose, we utilized cells transfected with plasmids encoding fluorescently tagged receptors, which were exposed to dcEF in a custom-designed electrotactic chamber. Fluorescent images were captured using wide-field or TIRF microscopy, enabling precise quantitative analysis of receptor redistribution. Additionally, the functional significance of these selected receptors in electrotaxis was evaluated by silencing their expression using an siRNA library.
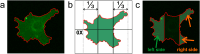
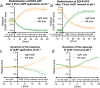
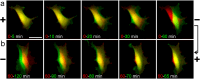
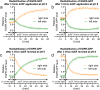
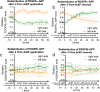
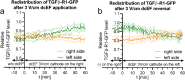
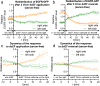
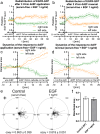
Results: Although EGFR moved immediately to cathode after dcEF application, maximum distribution asymmetry was reached after 30-40 min. This process was more efficient at higher dcEF intensities, specifically, asymmetry was greater at 3 V/cm compared to 1 V/cm, consistent with the biphasic mechanism observed only under the stronger dcEF. Additionally, redistribution was more effective under alkaline conditions and near the cell base, but decreased when glass was coated with poly-L-lysine, indicating electroosmosis as a key factor. Importantly, EGFR redistribution did not correlate with the rapid reaction of 3T3 cells to dcEF reversal, which occurred within 1-2 min, when receptor orientation was not yet reversed. PDGFRα exhibited similar but less marked cathodal redistribution, while PDGFRβ and TGFβR1 did not redistribute. siRNA knockdown experiments confirmed the importance of EGFR and ErbB4 in the electrotaxis. EGFR's role was largely ligand-independent, and it had a significant impact on the response of 3T3 cells to dcEF during the first hour of the experiment, but was not involved in the fastest response, which was Kir-dependent.
Conclusions: Our study suggests that EGFR redistribution may play a role in the early stages and partially contribute to the long-term electrotaxis of 3T3 fibroblasts. However, this mechanism alone does not fully explain rapid responses to dcEF orientation changes indicating a more complex, multimodal mechanism of electrotaxis in these cells.
Keywords: EGFR; Electric field; Electroosmosis; Electrotaxis; Receptor redistribution; dcEF.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures





Similar articles
-
The dynamics of the electrotactic reaction of mouse 3T3 fibroblasts.Biochim Biophys Acta Mol Cell Res. 2024 Feb;1871(2):119647. doi: 10.1016/j.bbamcr.2023.119647. Epub 2023 Dec 11. Biochim Biophys Acta Mol Cell Res. 2024. PMID: 38092134
-
Evaluation of EGFR and RTK signaling in the electrotaxis of lung adenocarcinoma cells under direct-current electric field stimulation.PLoS One. 2013 Aug 9;8(8):e73418. doi: 10.1371/journal.pone.0073418. eCollection 2013. PLoS One. 2013. PMID: 23951353 Free PMC article.
-
Control of neonatal human dermal fibroblast migration on poly(lactic-co-glycolic acid)-coated surfaces by electrotaxis.J Tissue Eng Regen Med. 2017 Mar;11(3):862-868. doi: 10.1002/term.1986. Epub 2015 Jan 28. J Tissue Eng Regen Med. 2017. PMID: 25627750
-
Recent Developments in Electrotaxis Assays.Adv Wound Care (New Rochelle). 2014 Feb 1;3(2):149-155. doi: 10.1089/wound.2013.0453. Adv Wound Care (New Rochelle). 2014. PMID: 24761355 Free PMC article. Review.
-
A microfluidic perspective on conventional in vitro transcranial direct current stimulation methods.J Neurosci Methods. 2023 Feb 1;385:109761. doi: 10.1016/j.jneumeth.2022.109761. Epub 2022 Dec 5. J Neurosci Methods. 2023. PMID: 36470469 Free PMC article. Review.
References
-
- McCaig CD, Song B, Rajnicek AM. Electrical dimensions in cell science. J Cell Sci. 2009;122:4267–76. 10.1242/jcs.023564. - PubMed
-
- Campetelli A, Bonazzi D, Minc N. Electrochemical regulation of cell Polarity and the cytoskeleton. Cytoskeleton. 2012;69:601–12. 10.1002/cm.21047. - PubMed
-
- Messerli MA, Graham DM. Extracellular electrical fields direct wound healing and regeneration. Biol Bull. 2011;221:79–92. 10.1086/BBLv221n1p79. - PubMed
-
- Nuccitelli R. A role for endogenous electric fields in wound healing. Curr Top Dev Biol. 2003;58:1–26. 10.1016/s0070-2153(03)58001-2. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

