Safety and Efficacy of a "High and Low Molecular Weight Hyaluronic Acid Hybrid Complex" Injection for Face Rejuvenation
- PMID: 40200860
- PMCID: PMC11979678
- DOI: 10.1111/jocd.70117
Safety and Efficacy of a "High and Low Molecular Weight Hyaluronic Acid Hybrid Complex" Injection for Face Rejuvenation
Abstract
Introduction: Hyaluronic acid (HA)-based formulations could have remarkable efficacy in treating a wide range of skin defects, including skin aging. The purpose of the study was the evaluation of the clinical safety and efficacy of a high and low molecular weight HA hybrid complex injection for skin rejuvenation.
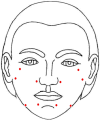
Method: In this single-arm, before-and-after clinical study, 20 subjects with wrinkled, dry, or rough skin were enrolled. They received two treatment sessions, each of 2 mL of stabilized high- and low-molecular-weight HA through intradermal injections in five bioaesthetic points with an interval of 4 weeks. Efficacy assessment measures included biophysical and sonographic parameters and the Global Aesthetic Improvement Scale (GAIS) score. Pain assessment, safety profile, and subject satisfaction were also reported.
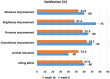
Results: A significant improvement in skin firmness was demonstrated in both follow-up visits. The Transepidermal water loss (TEWL) and the dermis ecodensity improved significantly in the first follow-up visit. A statistically significant increase in the dermis thickness was seen in the second follow-up visit. The median GAIS score indicated an average improvement of 51%-75%. The median overall satisfaction score was 7 and 6 in the first and second follow-up visits. No important side effects were observed. The average pain VAS score was 2 out of 10.
Conclusion: This new HA-based formulation is a safe and efficient treatment option to restore the vitality and turgidity of the skin.
Trial registration: ClinicalTrials.gov identifier: IRCT20150101020514N17 with ethics code IR.TUMS.TIPS.REC.1401.083.
Keywords: TEWL; dermis density; dermis thickness; elasticity; high and low molecular weight; hyaluronic acid; hydration.
© 2025 The Author(s). Journal of Cosmetic Dermatology published by Wiley Periodicals LLC.
Conflict of interest statement
Araz Sabzvari is a member of CinnaGen medical biotechnology research center, which collaborates with universities and researchers all over the world with regards to research and development of medications and health issues. Hamidreza Kafi is the head of the medical department of Orchid Pharmed Company which is in collaboration with Espad Pharmed Daru Company with respect to conducting clinical trials. Other authors declare no conflicts of interest.
Figures


References
-
- Dayan S., Rivkin A., Sykes J. M., et al., “Aesthetic Treatment Positively Impacts Social Perception: Analysis of Subjects From the HARMONY Study,” Aesthetic Surgery Journal 39 (2019): 1380–1389. - PubMed
-
- Voegeli R., Campiche R., Biassin R., Rawlings A. V., Shackelford T. K., and Fink B., “Predictors of Female Age, Health, and Attractiveness Perception From Skin Feature Analysis of Digital Portraits in Five Ethnic Groups,” International Journal of Cosmetic Science 45 (2023): 672–687. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

