Impact of genetic counseling and testing in individuals at high risk of familial Alzheimer's disease from Latin America: a non-randomized controlled trial
- PMID: 40201594
- PMCID: PMC11973255
- DOI: 10.1002/dad2.70102
Impact of genetic counseling and testing in individuals at high risk of familial Alzheimer's disease from Latin America: a non-randomized controlled trial
Abstract
Introduction: This study involved evaluating a tailored genetic counseling and testing (GCT) protocol for families at risk of autosomal dominant Alzheimer's disease (ADAD) in Latin America (LatAm), focusing on essential cultural and regional adaptations.
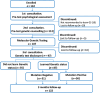
Methods: We conducted a non-randomized controlled trial among ADAD families in Colombia and Argentina. Participants were categorized based on their decision to learn their genetic status (GS), with further comparisons between mutation-positive versus mutation-negative participants who learned their status. Psychological impacts were measured using validated scales for anxiety and depression.
Results: Of the 122 eligible participants, 97 completed the GCT protocol, and 87 opted to learn their GS. There were no clinically significant differences in psychological distress between those who learned their status and those who did not, nor between mutation-positive and mutation-negative individuals.
Discussion: The GCT protocol effectively managed psychological impacts in ADAD families and was positively received, demonstrating the importance of culturally adapted GCT protocols.
Highlights: We examined the adaptation and efficacy of a GCT protocol in LatAm for families at risk of ADAD.The GCT protocol mitigated psychological distress among at-risk ADAD families.The study confirms the protocol's cultural appropriateness and psychological safety.Future studies should explore the long-term psychological and public health impacts of GCT and use of GCT for treatment options.
Keywords: Latin America; autosomal dominant Alzheimer's disease; cultural adaptation; genetic counseling and testing; psychological impact; vulnerabilities.
© 2025 The Author(s). Alzheimer's & Dementia: Diagnosis, Assessment & Disease Monitoring published by Wiley Periodicals, LLC on behalf of Alzheimer's Association.
Conflict of interest statement
The authors report no disclosures relevant to this manuscript. Author disclosures are available in the supporting information.
Figures


Similar articles
-
Nutritional interventions for survivors of childhood cancer.Cochrane Database Syst Rev. 2016 Aug 22;2016(8):CD009678. doi: 10.1002/14651858.CD009678.pub2. Cochrane Database Syst Rev. 2016. PMID: 27545902 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Psychological therapies for post-traumatic stress disorder and comorbid substance use disorder.Cochrane Database Syst Rev. 2016 Apr 4;4(4):CD010204. doi: 10.1002/14651858.CD010204.pub2. Cochrane Database Syst Rev. 2016. PMID: 27040448 Free PMC article.
-
Risk-stratified Care Improves Pain-related Knowledge and Reduces Psychological Distress for Low Back Pain: A Secondary Analysis of a Randomized Trial.Clin Orthop Relat Res. 2025 Apr 1;483(4):607-620. doi: 10.1097/CORR.0000000000003351. Epub 2025 Jan 21. Clin Orthop Relat Res. 2025. PMID: 39842027 Clinical Trial.
-
Pharmacological interventions for those who have sexually offended or are at risk of offending.Cochrane Database Syst Rev. 2015 Feb 18;2015(2):CD007989. doi: 10.1002/14651858.CD007989.pub2. Cochrane Database Syst Rev. 2015. PMID: 25692326 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
