Improvements induced by retinal gene therapy with voretigene neparvovec depend on visual cortical hemispheric dominance mechanisms
- PMID: 40204976
- PMCID: PMC11982196
- DOI: 10.1038/s43856-025-00820-y
Improvements induced by retinal gene therapy with voretigene neparvovec depend on visual cortical hemispheric dominance mechanisms
Abstract
Background: RPE65-associated retinal degeneration (RPE65-RD) causes severe visual deficits. Gene therapy with AAV2-hRPE65v2 is a breakthrough but it is currently unknown which visual pathways benefit from treatment and if cortical mechanisms can amplify retinal improvements.
Methods: In this within-subject design, ten patients with biallelic RPE65-RD underwent sub-retinal injection of AAV2-hRPE65v2. Psychophysical full-field stimulus threshold determination and functional magnetic resonance imaging were performed before and 12 months after treatment. Population receptive fields (pRF) were computed in V1 and visual responses assessed using contrast-reversed checkerboards (3 contrast levels).
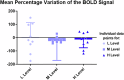
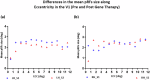
Results: Here we show significant improvement in light sensitivity at low-luminance and neural response enhancements under low-luminance conditions specifically in the right hemisphere, which is known to show dominance in attentional and visual pooling of spatial information. Changes in pRF size also reflect known hemispheric spatial asymmetries (left/right biased for local/global analysis, respectively).
Conclusions: Our findings show a contribution of known early and high-level cortical dominance mechanisms on improvement, which constrain the effects of therapy and are therefore a target for neurorehabilitation. These findings provide insight into the limits of clinical benefits of gene therapy and suggest that neurorehabilitation approaches may be needed to enhance improvements, similarly to cochlear implants.
Plain language summary
Retinal degeneration is caused by the loss of cells in the back of the eye and can lead to severe loss of vision. Here, we investigated the response of the brain to tests of vision in people who had been administered with gene therapy to treat retinal degeneration. Gene therapy is a treatment which aims to replace a gene that is not functioning correctly and thus causing disease. Ten patients were assessed before and a year after treatment using psychophysical tests and neuroimaging, and specific improvements in light sensitivity and neural responses were observed. The results suggest that rehabilitation approaches targeting the brain and vision may improve the results following this gene therapy.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures




Similar articles
-
Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65-mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial.Lancet. 2017 Aug 26;390(10097):849-860. doi: 10.1016/S0140-6736(17)31868-8. Epub 2017 Jul 14. Lancet. 2017. PMID: 28712537 Free PMC article. Clinical Trial.
-
Voretigene Neparvovec: A Review in RPE65 Mutation-Associated Inherited Retinal Dystrophy.Mol Diagn Ther. 2020 Aug;24(4):487-495. doi: 10.1007/s40291-020-00475-6. Mol Diagn Ther. 2020. PMID: 32535767 Review.
-
Single Center Experience with Voretigene Neparvovec Gene Augmentation Therapy in RPE65 Mutation-Associated Inherited Retinal Degeneration in a Clinical Setting.Ophthalmology. 2024 Feb;131(2):161-178. doi: 10.1016/j.ophtha.2023.09.006. Epub 2023 Sep 12. Ophthalmology. 2024. PMID: 37704110
-
Safety and durability of effect of contralateral-eye administration of AAV2 gene therapy in patients with childhood-onset blindness caused by RPE65 mutations: a follow-on phase 1 trial.Lancet. 2016 Aug 13;388(10045):661-72. doi: 10.1016/S0140-6736(16)30371-3. Epub 2016 Jun 30. Lancet. 2016. PMID: 27375040 Free PMC article. Clinical Trial.
-
Voretigene Neparvovec: An Emerging Gene Therapy for the Treatment of Inherited Blindness.2018 Mar 1. In: CADTH Issues in Emerging Health Technologies. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; 2016–2021. 169. 2018 Mar 1. In: CADTH Issues in Emerging Health Technologies. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; 2016–2021. 169. PMID: 30855774 Free Books & Documents. Review.
References
-
- den Hollander, A. I., Roepman, R., Koenekoop, R. K. & Cremers, F. P. M. Leber congenital amaurosis: genes, proteins and disease mechanisms. Prog. Retin. Eye Res.27, 391–419 (2008). - PubMed
-
- Mowat, F. M. et al. RPE65 gene therapy slows cone loss in Rpe65-deficient dogs. Gene Ther.20, 545–555 (2012). 2013 20:5. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

