Immune Checkpoint Inhibitors and Cardiovascular Adverse Events
- PMID: 40205958
- PMCID: PMC12287798
- DOI: 10.1002/ehf2.15281
Immune Checkpoint Inhibitors and Cardiovascular Adverse Events
Abstract
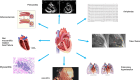
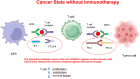
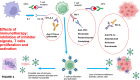
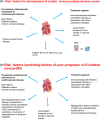
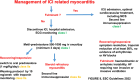
In the last years, we assisted to a tremendous increase in therapeutic options for the management of cancers, with immunotherapy at the forefront of this innovation. Immune checkpoint inhibitors (ICIs) have been developed to enhance the activity of the immune system against cancer cells (1) and the number of approvals for ICIs has rapidly increased. ICIs have also been associated with disinhibited cytotoxic T cells that damage healthy tissue in multiple organs, causing immune-related adverse events (AEs). Cardiovascular AEs (CVAe) are increasingly reported: myocarditis, Takotsubo syndrome, pericarditis and pericardial effusion, worsening of atherosclerosis, acute coronary syndromes, non-inflammatory heart failure, and ischaemic stroke. They are classified into five grades, based on presenting symptoms, level of cardiac biomarkers, and imaging. Even though myocarditis occurs more frequently than previously thought, clinically relevant myocarditis is a rare irAE compared to other irAE (0.5-1.2%). The clinical manifestations range from mild symptoms such as to chest pain, heart failure, and cardiogenic shock. The prognosis is severe, with mortality rates ranging from 25% to 50%. It is frequently associated with the concomitant use of combination of checkpoint inhibitors. The treatment strategies are tripartite: (i) holding ICI to prevent further toxicity, (ii) immunosuppression to alleviate inflammatory changes, and (iii) supportive therapy to address cardiac complications. Glucocorticoids represent the first-line treatment. In hemodynamically unstable patients, treatment with high-dose steroids should be initiated (intravenous methylprednisolone 1000 or 1250 mg oral methylprednisolone during 4 days). ICI-associated pericarditis can be accompanied by no/mild pericardial effusion up to cardiac tamponade. The treatment is made of nonsteroidal anti-inflammatory drugs and colchicine, corticosteroids if needed, and pericardiocentesis for the large effusions. ICIs could be continued for Grade 1 pericarditis, while temporary suspension of ICI is warranted for more severe cases. There is significant potential for accelerated atherosclerosis with ICIs as a long-term effect, but atherosclerosis-related CVAEs are not frequent, especially during treatment; increasing evidence associates ICIs with progression of atherosclerosis and increased atherosclerotic cardiovascular disease. ICIs can lead to arrhythmias: atrial fibrillation, supraventricular and ventricular tachycardias. Non-inflammatory heart failure syndrome have been observed in ICI-treated patients. Immune checkpoint inhibitors seem to be involved in the development of right ventricular dysfunction and pulmonary arterial hypertension. It is of the outmost importance to improve the collaboration among the different medical figures, such as cardiologists, oncologists, endocrinologists, and immunologists, both in clinical practice and in basic science research, to better recognize these adverse events, to understand their pathophysiological mechanisms, and to improve the overall survival and quality of life of the affected patients.
Keywords: Cardiovascular adverse events; Immune checkpoint inhibitors; Multidisciplinary decision making; Multimodality imaging; Myocarditis.
© 2025 The Author(s). ESC Heart Failure published by John Wiley & Sons Ltd on behalf of European Society of Cardiology.
Conflict of interest statement
G.M. has received advisory board or speaker's fees from AstraZeneca, Bayer, Boehringer Ingelheim, Daiichi Sankyo, Gebro Pharma, Novartis, and Vifor, all outside of the submitted work. M.L.D.P. received advisory board fee from Boehringer Ingelheim, outside of the submitted work. The other authors have no conflicts of interest.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

