Allosteric Differentiation of Al(I) Reactivity
- PMID: 40211470
- PMCID: PMC12117181
- DOI: 10.1002/chem.202501352
Allosteric Differentiation of Al(I) Reactivity
Abstract
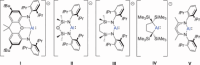
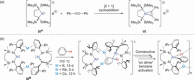
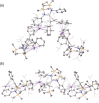
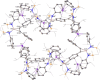
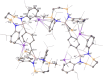
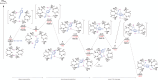
The dimeric potassium alumanyl, [{SiNDipp}AlK]2 ({SiNDipp} = {CH2SiMe2N(Dipp)}2; Dipp = 2,6-i-Pr2C6H3), reacts with two equivalents of PhC≡CR (R = Ph or SiMe3) with the exclusive formation of the aluminacyclopropene derivatives, [{SiNDipp}Al{η2-C2(R1)(R2)}]. In contrast, reactions with an equal stoichiometry of both alumanyl and alkyne reagents provide the products of not only alkyne cycloaddition but also para-C-H activation of a phenyl substituent. Supported by a theoretical study, this outcome is judged to result from a sequence of cooperative steps and the introduction of a modicum of kinetic discrimination that is suggested to be allosteric in origin.
Keywords: alkyne; aluminum; arene activation; cycloaddition; density functional theory.
© 2025 The Author(s). Chemistry – A European Journal published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Seven-Membered Cyclic Potassium Diamidoalumanyls.Chemistry. 2021 Oct 25;27(60):14971-14980. doi: 10.1002/chem.202102682. Epub 2021 Sep 21. Chemistry. 2021. PMID: 34403562 Free PMC article.
-
Alumanyl Reduction, Reductive Coupling and C-H Isomerization of Organic Nitriles.Organometallics. 2024 Aug 16;43(17):1938-1945. doi: 10.1021/acs.organomet.4c00289. eCollection 2024 Sep 9. Organometallics. 2024. PMID: 39268183 Free PMC article.
-
On the reactivity of Al-group 11 (Cu, Ag, Au) bonds.Dalton Trans. 2022 Mar 8;51(10):3913-3924. doi: 10.1039/d2dt00404f. Dalton Trans. 2022. PMID: 35169824
-
Aluminum-Boron Bond Formation by Boron Ester Oxidative Addition at an Alumanyl Anion.Inorg Chem. 2023 Sep 18;62(37):15310-15319. doi: 10.1021/acs.inorgchem.3c02566. Epub 2023 Sep 6. Inorg Chem. 2023. PMID: 37672789 Free PMC article.
-
Calcium-Catalyzed Arene C-H Bond Activation by Low-Valent AlI.Angew Chem Int Ed Engl. 2019 Oct 21;58(43):15496-15503. doi: 10.1002/anie.201908978. Epub 2019 Sep 13. Angew Chem Int Ed Engl. 2019. PMID: 31465144 Free PMC article. Review.
References
-
- (a) Motlagh H. N., Wrabl J. O., Li J., Hilser V. J., Nature 2014, 508, 331; - PMC - PubMed
- (b) Wodak S. J., Paci E., Dokholyan N. V., Berezovsky I. N., Horovitz A., Li J., Hilser V. J., Bahar I., Karanicolas J., Stock G., Hamm P., Stote R. H., Eberhardt J., Chebaro Y., Dejaegere A., Cecchini M., Changeux J. P., Bolhuis P. G., Vreede J., Faccioli P., Orioli S., Ravasio R., Yan L., Brito C., Wyart M., Gkeka P., Rivalta I., Palermo G., McCammon J. A., Panecka‐Hofman J., et al., Structure 2019, 27, 566; - PMC - PubMed
- (c) Liu J., Nussinov R., PLoS Comput. Biol. 2016, 12, e1004966; - PMC - PubMed
- (d) Fenton A. W., Trends Biochem. Sci. 2008, 33, 420. - PMC - PubMed
-
- (a) Rebek J., Trend J. E., Wattley R. V., Chakravorti S., J. Am. Chem. Soc. 1979, 101, 4333;
- (b) Rebek J., Wattley R. V., J. Am. Chem. Soc. 1980, 102, 4853;
- (c) Rebek J., Wattley R. V., Costello T., Gadwood R., Marshall L., Angew. Chem., Int. Ed. Engl. 1981, 20, 605;
- (d) Onan K., Rebek J., Costello T., Marshall L., J. Am. Chem. Soc. 1983, 105, 6759;
- (e) Rebek J., Marshall L., J. Am. Chem. Soc. 1983, 105, 6668;
- (f) J. Rebek, Jr. , Costello T., Marshall L., Wattley R., Gadwood R. C., Onan K., J. Am. Chem. Soc. 1985, 107, 7481;
- (g) Gavina F., Luis S. V., Costero A. M., Burguete M. I., Rebek J., J. Am. Chem. Soc. 1988, 110, 7140.
-
- Kremer C., Lützen A., Chem.‐Eur. J. 2013, 19, 6162. - PubMed
-
- (a) Gianneschi N. C., Bertin P. A., Nguyen S. T., Mirkin C. A., Zakharov L. N., Rheingold A. L., J. Am. Chem. Soc. 2003, 125, 10508; - PubMed
- (b) Gianneschi N. C., Masar M. S., Mirkin C. A., Acc. Chem. Res. 2005, 38, 825; - PubMed
- (c) Oliveri C. G., Ulmann P. A., Wiester M. J., Mirkin C. A., Acc. Chem. Res. 2008, 41, 1618; - PMC - PubMed
- (d) Yoon H. J., Kuwabara J., Kim J.‐H., Mirkin C. A., Science 2010, 330, 66; - PubMed
- (e) Wiester M. J., Ulmann P. A., Mirkin C. A., Angew. Chem., Int. Ed. 2011, 50, 114; - PubMed
- (f) McGuirk C. M., Mendez‐Arroyo J., Lifschitz A. M., Mirkin C. A., J. Am. Chem. Soc. 2014, 136, 16594; - PubMed
- (g) Lifschitz A. M., Rosen M. S., McGuirk C. M., Mirkin C. A., J. Am. Chem. Soc. 2015, 137, 7252; - PubMed
- (h) Lifschitz A. M., Young R. M., Mendez‐Arroyo J., Stern C. L., McGuirk C. M., Wasielewski M. R., Mirkin C. A., Nat. Commun. 2015, 6, 6541; - PMC - PubMed
- (i) Kovbasyuk L., Krämer R., Chem. Rev. 2004, 104, 3161. - PubMed
-
- (a) Dydio P., Rubay C., Gadzikwa T., Lutz M., Reek J. N. H., J. Am. Chem. Soc. 2011, 133, 17176; - PubMed
- (b) Mon I., Jose D. A., Vidal‐Ferran A., Chem.‐Eur. J. 2013, 19, 2720; - PubMed
- (c) McGuirk C. M., Stern C. L., Mirkin C. A., J. Am. Chem. Soc. 2014, 136, 4689; - PubMed
- (d) Marcos V., Stephens A. J., Jaramillo‐Garcia J., Nussbaumer A. L., Woltering S. L., Valero A., Lemonnier J. F., Vitorica‐Yrezabal I. J., Leigh D. A., Science 2016, 352, 1555; - PubMed
- (e) Kovbasyuk L., Pritzkow H., Krämer R., Fritsky I. O., Chem. Commun. 2004, 880; - PubMed
- (f) Smith A. J., Kalkman E. D., Gilbert Z. W., Tonks I. A., Organometallics 2016, 35, 2429;
- (g) Komon Z. J. A., Bu X. H., Bazan G. C., J. Am. Chem. Soc. 2000, 122, 12379;
- (h) Komon Z. J. A., Bu X. H., Bazan G. C., J. Am. Chem. Soc. 2000, 122, 1830;
- (i) Komon Z. J. A., Bazan G. C., Fang C., Bu X. H., Inorg. Chim. Acta 2003, 345, 95.
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

