Osteochondral tissue engineering‑based subchondral bone plate repair (Review)
- PMID: 40211705
- PMCID: PMC11997743
- DOI: 10.3892/mmr.2025.13517
Osteochondral tissue engineering‑based subchondral bone plate repair (Review)
Abstract
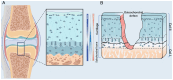
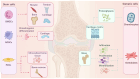
Osteochondral defects are a series of pathological changes from the chondral surface to the deeper trabecular bone caused by trauma or degenerative changes; they typically induce serious joint dysfunction. Over the past few decades, various techniques have been attempted to repair these defects. Tissue‑engineered osteochondral grafts (TEOGs) with sophisticated architecture have been extensively explored for osteochondral regeneration. However, controversies persist regarding standards for clinical application of TEOGs. The present review focused on the design of TEOGs, emphasizing their capacity to repair the subchondral bone plate (SBP). The effect of animal models on techniques to repair osteochondral defects was also reviewed. To improve the evaluation of SBP regeneration, four typical histological characteristics (abnormal height, uneven surface, poor integration and loose internal structure) are summarized based on cases of unsatisfactory SBP regeneration. Incorporating mesenchymal stem cells with appropriate growth factors into trilayer or multilayer tissue‑engineered scaffolds is a promising strategy to avoid unsatisfactory SBP regeneration. Large animal models are recommended for translation to the clinic and there is a need to establish detailed and comprehensive osteochondral defect models in the future.
Keywords: osteochondral defect; osteochondral regeneration; subchondral bone plate; tissue engineering strategy; translational animal.
Conflict of interest statement
The authors declare that they have no competing interests
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

