Chemical Vapor Deposition Mediated Phase Engineering for 2D Transition Metal Dichalcogenides: Strategies and Applications
- PMID: 40212328
- PMCID: PMC11935802
- DOI: 10.1002/smsc.202100047
Chemical Vapor Deposition Mediated Phase Engineering for 2D Transition Metal Dichalcogenides: Strategies and Applications
Abstract
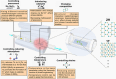
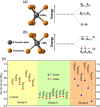
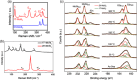
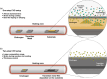
2D transition metal dichalcogenides (TMDs) are characterized by the presence of multiple crystal structures or phases, even at the ultrathin limit. Controlling phase transformations, namely, phase engineering, of 2D TMDs is crucial for realizing high-performance 2D devices by combining phases with distinct physical and chemical properties. As a powerful approach for large-scale production of high-quality 2D TMDs, chemical vapor deposition (CVD) offers unique advantages in phase engineering due to its highly controllable synthesis processes. Starting with an introduction of the crystal structures and phase transformations of 2D TMDs, this review summarizes the recent developments in CVD-mediated phase engineering strategies of TMDs, including control of temperature, precursors, catalysis, atmosphere, composition, and strain during the deposition process. Moreover, the representative applications of CVD-based phase-engineered TMDs in the field of transistors, photodetectors, photovoltaic cells, and catalysis are overviewed. Finally, the challenges, expectations of CVD-based phase engineering, and future development of this versatile technique are discussed.
Keywords: 2D transition metal dichalcogenides; chemical vapor deposition; phase-selective growth.
© 2021 The Authors. Small Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures













Similar articles
-
Substrate Engineering for Chemical Vapor Deposition Growth of Large-Scale 2D Transition Metal Dichalcogenides.Adv Mater. 2023 Dec;35(52):e2211855. doi: 10.1002/adma.202211855. Epub 2023 Nov 2. Adv Mater. 2023. PMID: 37095721 Review.
-
Recent Advances in Spin-coating Precursor Mediated Chemical Vapor Deposition of Two-Dimensional Transition Metal Dichalcogenides.Precis Chem. 2024 Feb 13;2(7):282-299. doi: 10.1021/prechem.3c00115. eCollection 2024 Jul 22. Precis Chem. 2024. PMID: 39473898 Free PMC article. Review.
-
Recent Progress in CVD Growth of 2D Transition Metal Dichalcogenides and Related Heterostructures.Adv Mater. 2019 Oct;31(41):e1901694. doi: 10.1002/adma.201901694. Epub 2019 Aug 12. Adv Mater. 2019. PMID: 31402526 Review.
-
Strategies for Controlled Growth of Transition Metal Dichalcogenides by Chemical Vapor Deposition for Integrated Electronics.ACS Mater Au. 2022 Jul 8;2(6):665-685. doi: 10.1021/acsmaterialsau.2c00029. eCollection 2022 Nov 9. ACS Mater Au. 2022. PMID: 36855548 Free PMC article. Review.
-
Recent Developments in Controlled Vapor-Phase Growth of 2D Group 6 Transition Metal Dichalcogenides.Adv Mater. 2019 May;31(20):e1804939. doi: 10.1002/adma.201804939. Epub 2019 Feb 1. Adv Mater. 2019. PMID: 30706541 Review.
Cited by
-
A Review on Optical Biosensors for Monitoring of Uric Acid and Blood Glucose Using Portable POCT Devices: Status, Challenges, and Future Horizons.Biosensors (Basel). 2025 Mar 31;15(4):222. doi: 10.3390/bios15040222. Biosensors (Basel). 2025. PMID: 40277536 Free PMC article. Review.
-
Stoichiometry-engineered phase transition in a two-dimensional binary compound.Nat Commun. 2025 May 5;16(1):4162. doi: 10.1038/s41467-025-59429-3. Nat Commun. 2025. PMID: 40324982 Free PMC article.
References
-
- Novoselov K. S., Geim A. K., Morozov S. V., Jiang D., Zhang Y., Dubonos S. V., Grigorieva I. V., Firsov A. A., Science 2004, 306, 666. - PubMed
-
- Mas-Ballesté R., Gómez-Navarro C., Gómez-Herrero J., Zamora F., Nanoscale 2011, 3, 20. - PubMed
-
- a) Bertolazzi S., Bondavalli P., Roche S., San T., Choi S.-Y., Colombo L., Bonaccorso F., Samorì P., Adv. Mater. 2019, 31, 1806663; - PubMed
- b) Xia F., Wang H., Xiao D., Dubey M., Ramasubramaniam A., Nat. Photonics 2014, 8, 899;
- c) Liang S.-J., Cheng B., Cui X., Miao F., Adv. Mater. 2020, 32, 1903800; - PubMed
- d) Zhu W., Low T., Wang H., Ye P., Duan X., 2D Mater. 2019, 6, 032004;
- e) Meng C., Das P., Shi X., Fu Q., Müllen K., Wu Z.-S., Small Sci. 2021, 1, 2000076.
-
- Kappera R., Voiry D., Yalcin S. E., Branch B., Gupta G., Mohite A. D., Chhowalla M., Nat. Mater. 2014, 13, 1128. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
