Functionalization of Silica Nanoparticles for Tailored Interactions with Intestinal Cells and Chemical Modulation of Paracellular Permeability
- PMID: 40212655
- PMCID: PMC11934983
- DOI: 10.1002/smsc.202400112
Functionalization of Silica Nanoparticles for Tailored Interactions with Intestinal Cells and Chemical Modulation of Paracellular Permeability
Abstract
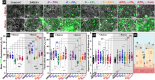
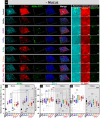
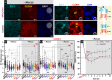
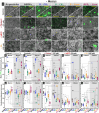
The intestinal compartment confines the gut microbiome while enabling food passage and absorption of active molecules. For the rational design of oral formulations aiming to overcome physiological barriers of the gut, it is crucial to understand how cells respond to the presence of nanoparticulate materials. Taking advantage of the versatility and biocompatibility of dendritic mesoporous silica nanoparticles (DMSNs), several post-grafting strategies are developed to diversify the surface properties of spherical DMSNs and then probe interactions with the intestinal coculture cell model Caco-2/HT29-MTX-E12. Herein, the functionalization of DMSNs with polyethylene glycol, phosphonate, methyl, and farnesol moieties enables the investigation of both particle penetration through the mucus layer and pathways relevant to intracellular uptake. Contributions of surface chemistry, charge, and colloidal stability are correlated with the modulation of particle movement through the mucus and the organization of cell-cell junctions. Hydrophilic and negative functionalities favor particle distribution toward the intestinal monolayer. Instead, hydrophobic DMSNs are hindered by the mucus, possibly limiting cell contact. Hybrid surfaces, combining phosphonate and long carbon chain functions, support diffusion through the mucus and foster the paracellular permeability as well as the transient barrier relapse, as indicated by increased cell-cell distances and reorganization of tight junctions.
Keywords: differentiated intestinal cells; membrane permeation; mesoporous silica nanoparticles; mucus barrier; surface functions; tight junction proteins.
© 2024 The Author(s). Small Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Morphology-Dependent Interaction of Silica Nanoparticles with Intestinal Cells: Connecting Shape to Barrier Function.Nano Lett. 2023 Aug 23;23(16):7758-7766. doi: 10.1021/acs.nanolett.3c00835. Epub 2023 Jul 11. Nano Lett. 2023. PMID: 37433061 Free PMC article.
-
Small silica nanoparticles transiently modulate the intestinal permeability by actin cytoskeleton disruption in both Caco-2 and Caco-2/HT29-MTX models.Arch Toxicol. 2020 Apr;94(4):1191-1202. doi: 10.1007/s00204-020-02694-6. Epub 2020 Mar 11. Arch Toxicol. 2020. PMID: 32162006
-
Mucus-penetrating dendritic mesoporous silica nanoparticle loading drug nanocrystal clusters to enhance permeation and intestinal absorption.Biomater Sci. 2023 Jan 31;11(3):1013-1030. doi: 10.1039/d2bm01404a. Biomater Sci. 2023. PMID: 36545798
-
Enteric Pathogens and Their Toxin-Induced Disruption of the Intestinal Barrier through Alteration of Tight Junctions in Chickens.Toxins (Basel). 2017 Feb 10;9(2):60. doi: 10.3390/toxins9020060. Toxins (Basel). 2017. PMID: 28208612 Free PMC article. Review.
-
The Caco-2 Model: Modifications and enhancements to improve efficiency and predictive performance.Int J Pharm. 2022 Aug 25;624:122004. doi: 10.1016/j.ijpharm.2022.122004. Epub 2022 Jul 9. Int J Pharm. 2022. PMID: 35820514 Review.
References
LinkOut - more resources
Full Text Sources
