Drosophila AHR limits tumor growth and stem cell proliferation in the intestine
- PMID: 40212817
- PMCID: PMC11982807
- DOI: 10.12688/wellcomeopenres.23515.3
Drosophila AHR limits tumor growth and stem cell proliferation in the intestine
Abstract
Background: The aryl hydrocarbon receptor (AHR) plays important roles in intestinal homeostasis, limiting tumour growth and promoting differentiation in the intestinal epithelium. Spineless, the Drosophila homolog of AHR, has only been studied in the context of development but not in the adult intestine.
Methods: The role of Spineless in the Drosophila midgut was studied by overexpression or inactivation of Spineless in infection and tumour models and RNA sequencing of sorted midgut progenitor cells.
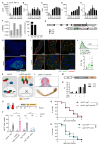
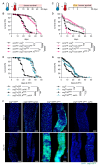
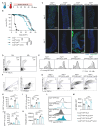
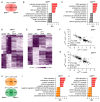
Results: We show that spineless is upregulated in the adult intestinal epithelium after infection with Pseudomonas entomophila ( P. e.). Spineless inactivation increased stem cell proliferation following infection-induced injury. Spineless overexpression limited intestinal stem cell proliferation and reduced survival after infection. In two tumour models, using either Notch RNAi or constitutively active Yorkie, Spineless suppressed tumour growth and doubled the lifespan of tumour-bearing flies. At the transcriptional level it reversed the gene expression changes induced in Yorkie tumours, counteracting cell proliferation and altered metabolism.
Conclusions: These findings demonstrate a new role for Spineless in the adult Drosophila midgut and highlight the evolutionarily conserved functions of AHR/Spineless in the control of proliferation and differentiation of the intestinal epithelium.
Keywords: Drosophila; aryl hydrocarbon receptor; intestinal epithelium; spineless; tumour suppressor.
Plain language summary
The transcription factor aryl hydrocarbon receptor (AHR) plays important roles in the intestine, limiting tumour growth and promoting the normal epithelial lining. Spineless, the fruit fly homolog of AHR, has only been studied in the context of embryonic development but not in the intestine of adult flies. Here, we show that spineless is upregulated in the adult intestinal epithelium after infection with a bacterium. Blocking Spineless function increased stem cell proliferation after bacterial infection. Increasing Spineless had the opposite effect and limited intestinal stem cell proliferation. It also reduced survival after bacterial infection. In two separate tumour models, Spineless suppressed tumour growth and doubled the lifespan of tumour-bearing flies. Increasing Spineless reversed the gene expression changes induced in tumours, counteracting cell proliferation and changes to the cellular metabolism. These findings demonstrate a new role for Spineless in the adult fruit fly midgut and highlight the evolutionarily conserved functions of mammalian AHR and fruit fly Spineless in the control of proliferation and differentiation of the intestinal epithelium.
Copyright: © 2025 Tsai M et al.
Conflict of interest statement
No competing interests were disclosed.
Figures



Similar articles
-
The spineless-aristapedia and tango bHLH-PAS proteins interact to control antennal and tarsal development in Drosophila.Development. 1999 Sep;126(17):3937-45. doi: 10.1242/dev.126.17.3937. Development. 1999. PMID: 10433921
-
Combination of hypomorphic mutations of the Drosophila homologues of aryl hydrocarbon receptor and nucleosome assembly protein family genes disrupts morphogenesis, memory and detoxification.PLoS One. 2014 Apr 15;9(4):e94975. doi: 10.1371/journal.pone.0094975. eCollection 2014. PLoS One. 2014. PMID: 24736732 Free PMC article.
-
Transcriptional Regulation of Math1 by Aryl Hydrocarbon Receptor: Effect on Math1+ Progenitor Cells in Mouse Small Intestine.Mol Cell Biol. 2023 Jan;43(1):43-63. doi: 10.1080/10985549.2022.2160610. Epub 2023 Jan 26. Mol Cell Biol. 2023. PMID: 36720468 Free PMC article.
-
Aryl hydrocarbon receptor (AhR) and pregnane X receptor (PXR) play both distinct and common roles in the regulation of colon homeostasis and intestinal carcinogenesis.Biochem Pharmacol. 2023 Oct;216:115797. doi: 10.1016/j.bcp.2023.115797. Epub 2023 Sep 9. Biochem Pharmacol. 2023. PMID: 37696457 Review.
-
Intestinal stem cell proliferation and epithelial homeostasis in the adult Drosophila midgut.Insect Biochem Mol Biol. 2015 Dec;67:9-14. doi: 10.1016/j.ibmb.2015.05.016. Epub 2015 May 27. Insect Biochem Mol Biol. 2015. PMID: 26024801 Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

