Dynamic gamma modulation of hippocampal place cells predominates development of theta sequences
- PMID: 40213917
- PMCID: PMC11991698
- DOI: 10.7554/eLife.97334
Dynamic gamma modulation of hippocampal place cells predominates development of theta sequences
Abstract
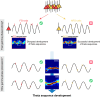
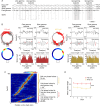
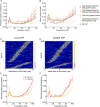
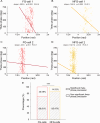
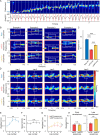
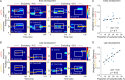
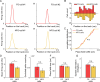
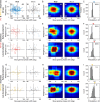
The experience-dependent spatial cognitive process requires sequential organization of hippocampal neural activities by theta rhythm, which develops to represent highly compressed information for rapid learning. However, how the theta sequences were developed in a finer timescale within theta cycles remains unclear. In this study, we found in rats that sweep-ahead structure of theta sequences developing with exploration was predominantly dependent on a relatively large proportion of FG-cells, that is a subset of place cells dominantly phase-locked to fast gamma rhythms. These ensembles integrated compressed spatial information by cells consistently firing at precessing slow gamma phases within the theta cycle. Accordingly, the sweep-ahead structure of FG-cell sequences was positively correlated with the intensity of slow gamma phase precession, in particular during early development of theta sequences. These findings highlight the dynamic network modulation by fast and slow gamma in the development of theta sequences which may further facilitate memory encoding and retrieval.
Keywords: gamma rhythms; hippocampus; neuroscience; phase precession; place cells; rat; theta sequence.
© 2024, Wang et al.
Conflict of interest statement
NW, YW, MG, LW, XW, NZ, JY, LW, CZ, DM No competing interests declared
Figures




Update of
- doi: 10.1101/2024.03.27.586908
- doi: 10.7554/eLife.97334.1
- doi: 10.7554/eLife.97334.2
- doi: 10.7554/eLife.97334.3
Similar articles
-
Spatial Sequence Coding Differs during Slow and Fast Gamma Rhythms in the Hippocampus.Neuron. 2016 Jan 20;89(2):398-408. doi: 10.1016/j.neuron.2015.12.005. Epub 2016 Jan 7. Neuron. 2016. PMID: 26774162 Free PMC article.
-
Coordinated Emergence of Hippocampal Replay and Theta Sequences during Post-natal Development.Curr Biol. 2019 Mar 4;29(5):834-840.e4. doi: 10.1016/j.cub.2019.01.005. Epub 2019 Feb 14. Curr Biol. 2019. PMID: 30773370 Free PMC article.
-
Hippocampal place cell sequences differ during correct and error trials in a spatial memory task.Nat Commun. 2021 Jun 7;12(1):3373. doi: 10.1038/s41467-021-23765-x. Nat Commun. 2021. PMID: 34099727 Free PMC article.
-
Theta-gamma coupling in the entorhinal-hippocampal system.Curr Opin Neurobiol. 2015 Apr;31:45-50. doi: 10.1016/j.conb.2014.08.001. Epub 2014 Aug 27. Curr Opin Neurobiol. 2015. PMID: 25168855 Free PMC article. Review.
-
Theta phase precession beyond the hippocampus.Rev Neurosci. 2012;23(1):39-65. doi: 10.1515/revneuro-2011-0064. Rev Neurosci. 2012. PMID: 22718612 Review.
References
MeSH terms
Grants and funding
- 2022ZD0205000/National Science and Technology Innovation 2030 Major Project of China
- T2322021/National Natural Science Foundation of China
- 82271218/National Natural Science Foundation of China
- 12271272/National Natural Science Foundation of China
- 81925020/National Natural Science Foundation of China
LinkOut - more resources
Full Text Sources

