MIRO1 Is Required for Dynamic Increases in Mitochondria-ER Contact Sites and Mitochondrial ATP During the Cell Cycle
- PMID: 40214436
- PMCID: PMC11988184
- DOI: 10.3390/cells14070482
MIRO1 Is Required for Dynamic Increases in Mitochondria-ER Contact Sites and Mitochondrial ATP During the Cell Cycle
Abstract
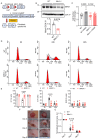
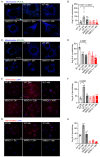
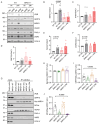
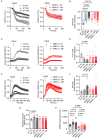
Mitochondria-ER contact sites (MERCS) are vital for mitochondrial dynamics, lipid exchange, Ca2+ homeostasis, and energy metabolism. We examined whether mitochondrial metabolism changes during the cell cycle depend on MERCS dynamics and are regulated by the outer mitochondrial protein mitochondrial rho GTPase 1 (MIRO1). Wound healing was assessed in mice with fibroblast-specific deletion of MIRO1. Wild-type and MIRO1-/- fibroblasts and vascular smooth muscle cells were evaluated for proliferation, cell cycle progression, number of MERCS, distance, and protein composition throughout the cell cycle. Restoration of MIRO1 mutants was used to test the role of MIRO1 domains; Ca2+ transients and mitochondrial metabolism were evaluated using biochemical, immunodetection, and fluorescence techniques. MERCS increased in number during G1/S compared with during G0, which was accompanied by a notable rise in protein-protein interactions involving VDAC1 and IP3R as well as GRP75 and MIRO1 by proximity-ligation assays. Split-GFP ER/mitochondrial contacts of 40 nm also increased. Mitochondrial Ca2+ concentration ([Ca2+]), membrane potential, and ATP levels correlated with the formation of MERCS during the cell cycle. MIRO1 deficiency blocked G1/S progression and the cell-cycle-dependent formation of MERCS and altered ER Ca2+ release and mitochondrial Ca2+ uptake. MIRO1 mutants lacking the Ca2+-sensitive EF hands or the transmembrane domain did not rescue cell proliferation or the formation of MERCS. MIRO1 controls an increase in the number of MERCS during cell cycle progression and increases mitochondrial [Ca2+], driving metabolic activity and proliferation through its EF hands.
Keywords: Ca2+; ER; MAM; MERCS; MIRO1; cell cycle; fibroblasts; mitochondria; vascular smooth muscle cells.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
Impaired mitochondrial-endoplasmic reticulum interaction and mitophagy in Miro1-mutant neurons in Parkinson's disease.Hum Mol Genet. 2020 May 28;29(8):1353-1364. doi: 10.1093/hmg/ddaa066. Hum Mol Genet. 2020. PMID: 32280985 Free PMC article.
-
Regulation of mitochondrial cristae organization by Myo19, Miro1 and Miro2, and metaxin 3.J Cell Sci. 2025 May 1;138(9):JCS263637. doi: 10.1242/jcs.263637. Epub 2025 Mar 6. J Cell Sci. 2025. PMID: 39882711 Free PMC article.
-
Mutations in RHOT1 Disrupt Endoplasmic Reticulum-Mitochondria Contact Sites Interfering with Calcium Homeostasis and Mitochondrial Dynamics in Parkinson's Disease.Antioxid Redox Signal. 2019 Dec 1;31(16):1213-1234. doi: 10.1089/ars.2018.7718. Epub 2019 Aug 21. Antioxid Redox Signal. 2019. PMID: 31303019 Free PMC article.
-
Ca2+ handling at the mitochondria-ER contact sites in neurodegeneration.Cell Calcium. 2021 Sep;98:102453. doi: 10.1016/j.ceca.2021.102453. Epub 2021 Aug 5. Cell Calcium. 2021. PMID: 34399235 Review.
-
The ER-mitochondria Ca2+ signaling in cancer progression: Fueling the monster.Int Rev Cell Mol Biol. 2021;363:49-121. doi: 10.1016/bs.ircmb.2021.03.006. Epub 2021 Apr 12. Int Rev Cell Mol Biol. 2021. PMID: 34392932 Review.
Cited by
-
MIRO1 controls energy production and proliferation of vascular smooth muscle cells.bioRxiv [Preprint]. 2025 Jun 9:2024.08.13.607854. doi: 10.1101/2024.08.13.607854. bioRxiv. 2025. PMID: 39185180 Free PMC article. Preprint.
References
-
- Ardail D., Popa I., Bodennec J., Louisot P., Schmitt D., Portoukalian J. The mitochondria-associated endoplasmic-reticulum subcompartment (MAM fraction) of rat liver contains highly active sphingolipid-specific glycosyltransferases. Biochem. J. 2003;371:1013–1019. doi: 10.1042/BJ20021834. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

