Prostate-Specific Membrane Antigen-Targeted Antibody-Drug Conjugates: A Promising Approach for Metastatic Castration-Resistant Prostate Cancer
- PMID: 40214467
- PMCID: PMC11989178
- DOI: 10.3390/cells14070513
Prostate-Specific Membrane Antigen-Targeted Antibody-Drug Conjugates: A Promising Approach for Metastatic Castration-Resistant Prostate Cancer
Abstract
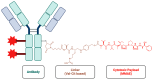
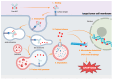
Prostate cancer (PCa), especially metastatic castration-resistant prostate cancer (mCRPC), is a significant cancer characterized by its poor prognosis and limited treatment options. Prostate-specific membrane antigen (PSMA) has emerged as a diagnostic and therapeutic target for PCa due to its restricted expression in malignant prostate tissues. In this case, several PSMA-targeting molecules were developed for radiotherapy and immunotherapy. Antibody-drug conjugates (ADCs) are a novel therapeutic approach for various carcinomas that can selectively target PSMA-positive tumor cells and minimize off-target toxicity. ADCs have made great progress in the treatment of breast and bladder cancers, and some have received FDA approval for target therapy. However, studies on PSMA ADCs are limited, and most clinical trials are at stage I or II. Therefore, this study reviewed trials about PSMA-targeting ADCs for the treatment of PCa. Clinical trials have reported a favorable pharmacokinetic profile and antitumor activity. Toxicity studies have revealed manageable adverse effects, with no significant off-target toxicity in PSMA-negative tissues. This study highlights the therapeutic potential of PSMA ADCs for the treatment of mCRPC. However, it also emphasizes the necessity of further clinical investigation to optimize efficacy, safety, and patient outcomes.
Keywords: chemotherapy; metastasis; prostate neoplasm; systemic therapy; target therapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Preclinical Characterization of ARX517, a Site-Specific Stable PSMA-Targeted Antibody-Drug Conjugate for the Treatment of Metastatic Castration-Resistant Prostate Cancer.Mol Cancer Ther. 2024 Dec 3;23(12):1842-1853. doi: 10.1158/1535-7163.MCT-23-0927. Mol Cancer Ther. 2024. PMID: 39172730 Free PMC article.
-
Efficacy Against Human Prostate Cancer by Prostate-specific Membrane Antigen-specific, Transforming Growth Factor-β Insensitive Genetically Targeted CD8+ T-cells Derived from Patients with Metastatic Castrate-resistant Disease.Eur Urol. 2018 May;73(5):648-652. doi: 10.1016/j.eururo.2017.12.008. Epub 2017 Dec 21. Eur Urol. 2018. PMID: 29275833 Free PMC article. Review.
-
Development of 5D3-DM1: A Novel Anti-Prostate-Specific Membrane Antigen Antibody-Drug Conjugate for PSMA-Positive Prostate Cancer Therapy.Mol Pharm. 2020 Sep 8;17(9):3392-3402. doi: 10.1021/acs.molpharmaceut.0c00457. Epub 2020 Aug 17. Mol Pharm. 2020. PMID: 32803984 Free PMC article.
-
The PSMA-targeting Half-life Extended BiTE Therapy AMG 160 has Potent Antitumor Activity in Preclinical Models of Metastatic Castration-resistant Prostate Cancer.Clin Cancer Res. 2021 May 15;27(10):2928-2937. doi: 10.1158/1078-0432.CCR-20-3725. Epub 2021 Jan 27. Clin Cancer Res. 2021. PMID: 33504551
-
Prostate-Specific Membrane Antigen-Directed Therapy for Metastatic Castration-Resistant Prostate Cancer.Cancer J. 2016 Sep/Oct;22(5):347-352. doi: 10.1097/PPO.0000000000000221. Cancer J. 2016. PMID: 27749329 Free PMC article. Review.
References
-
- Francini E., Gray K.P., Shaw G.K., Evan C.P., Hamid A.A., Perry C.E., Kantoff P.W., Taplin M.E., Sweeney C.J. Impact of new systemic therapies on overall survival of patients with metastatic castration-resistant prostate cancer in a hospital-based registry. Prostate Cancer Prostatic Dis. 2019;22:420–427. doi: 10.1038/s41391-018-0121-2. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

