The Ceramide-Dependent EV Secretome Differentially Affects Prostate Cancer Cell Migration
- PMID: 40214501
- PMCID: PMC11988362
- DOI: 10.3390/cells14070547
The Ceramide-Dependent EV Secretome Differentially Affects Prostate Cancer Cell Migration
Abstract
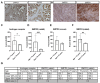
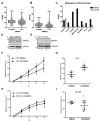
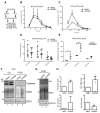
Tumor-derived extracellular vesicles (EVs) play an important role in cancer progression. Neutral sphingomyelinases (nSMases) are lipid-modifying enzymes that modulate the secretion of EVs from cells. How nSMase activity and therefore ceramide generation affect the composition and functionality of secreted EVs is not fully understood. Here, we aimed to investigate the expression of nSMases 1 and 2 in prostate cancer (PCa) tissue and their role in EV composition and secretion for prostate cancer cell migration. Reduced nSMase 1 and 2 expression was found in prostate cancer and correlated with the age of the patient. When nSMase 2 was inhibited by GW4869 in PCa cells (PC3 and DU145), the EV secretome was significantly altered, while the number of EVs and the total protein content of released EVs were not significantly changed. Using proteomic analysis, we found that extracellular matrix proteins, such as SDC4 (Syndecan-4) and SRPX-2, were differentially secreted on EVs from GW4869-treated PC3 cells. In scratch wound migration assays, GW4869 significantly increased migration compared to control PC3 cells but not DU145 cells, while SDC4 knockdown significantly reduced the migration of PC3 cells. These and other nSMase-2-dependent secreted proteins are interesting candidates for understanding the role of stress-induced EVs in the progression of prostate cancer.
Keywords: SMPD2; SMPD3; cell migration; extracellular matrix; neutral sphingomyelinases.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Casanova-Salas I., Aguilar D., Cordoba-Terreros S., Agundez L., Brandariz J., Herranz N., Mas A., Gonzalez M., Morales-Barrera R., Sierra A., et al. Circulating tumor extracellular vesicles to monitor metastatic prostate cancer genomics and transcriptomic evolution. Cancer Cell. 2024;42:1301–1312.e7. doi: 10.1016/j.ccell.2024.06.003. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

