Synthesis and Characterization of a Novel Sol-Gel-Derived Ni-Doped TiO2 Photocatalyst for Rapid Visible Light-Driven Mineralization of Paracetamol
- PMID: 40214575
- PMCID: PMC11990163
- DOI: 10.3390/nano15070530
Synthesis and Characterization of a Novel Sol-Gel-Derived Ni-Doped TiO2 Photocatalyst for Rapid Visible Light-Driven Mineralization of Paracetamol
Abstract
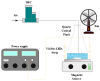
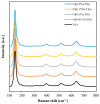
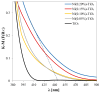
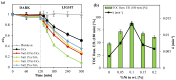
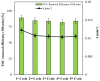
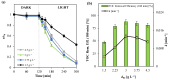
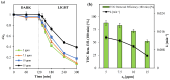
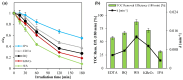
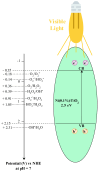
The increasing presence of pharmaceutical contaminants, such as paracetamol, in water sources necessitates the development of efficient and sustainable treatment technologies. This study investigates the photocatalytic degradation and mineralization of paracetamol under visible light using nickel-doped titanium dioxide (Ni-TiO2) catalysts synthesized via the sol-gel method. The catalysts were characterized through Raman spectroscopy, UV-Vis diffuse reflectance spectroscopy (UV-Vis DRS), and surface area measurements. Ni doping enhanced the visible light absorption of TiO2, reducing its band gap from 3.11 eV (undoped) to 2.49 eV at 0.20 wt.% Ni loading, while Raman analysis confirmed Ni incorporation with anatase as the predominant phase. The Ni(0.1%)-TiO2 catalyst exhibited the highest photocatalytic activity, achieving 88% total organic carbon (TOC) removal of paracetamol (5 ppm) after 180 min under optimal conditions (catalyst dosage, 3 g L-1). Stability tests demonstrated 84% retained efficiency over five cycles, with a kinetic rate constant of 0.010 min-1. Hydroxyl radicals were identified as the main reactive species. The catalyst maintained high performance in tap water, achieving 78.8% TOC removal. These findings highlight the potential of Ni(0.1%)-TiO2 as a cost-effective, visible light-active photocatalyst for the removal of pharmaceutical pollutants, with promising scalability for industrial water treatment applications.
Keywords: Ni-TiO2; catalyst stability; heterogeneous photocatalysis; paracetamol; pharmaceutical pollutants; slurry photoreactor; sol-gel synthesis; visible light driven.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Visible Light-Driven Photocatalytic Activity and Kinetics of Fe-Doped TiO2 Prepared by a Three-Block Copolymer Templating Approach.Materials (Basel). 2021 Jun 5;14(11):3105. doi: 10.3390/ma14113105. Materials (Basel). 2021. PMID: 34198890 Free PMC article.
-
Graphene oxide/titania photocatalytic ozonation of primidone in a visible LED photoreactor.J Hazard Mater. 2019 May 5;369:70-78. doi: 10.1016/j.jhazmat.2019.02.025. Epub 2019 Feb 10. J Hazard Mater. 2019. PMID: 30772689
-
Photocatalytic Disinfection of Selected Waterborne Pathogens by Visible Light-Active Nano Iron-Doped TiO2 Obtained by a Sol-Gel Method.ACS Appl Nano Mater. 2025 Apr 9;8(19):10066-10079. doi: 10.1021/acsanm.5c01408. eCollection 2025 May 16. ACS Appl Nano Mater. 2025. PMID: 40400699 Free PMC article.
-
Progress in photocatalytic degradation of industrial organic dye by utilising the silver doped titanium dioxide nanocomposite.Heliyon. 2024 Dec 5;10(24):e40998. doi: 10.1016/j.heliyon.2024.e40998. eCollection 2024 Dec 30. Heliyon. 2024. PMID: 39720083 Free PMC article. Review.
-
A comprehensive appraisal on status and management of remediation of DBPs by TiO2 based-photocatalysts: Insights of technology, performance and energy efficiency.J Environ Manage. 2023 Feb 15;328:117011. doi: 10.1016/j.jenvman.2022.117011. Epub 2022 Dec 14. J Environ Manage. 2023. PMID: 36525732 Review.
Cited by
-
Harnessing BiOI/V2O5 Nanocomposites: Advanced Bifunctional Catalysts for Visible-Light Driven Environmental Remediation and Antibacterial Activity.Molecules. 2025 Jun 6;30(12):2500. doi: 10.3390/molecules30122500. Molecules. 2025. PMID: 40572466 Free PMC article.
References
-
- Peralta-Hernández J.M., Brillas E. A Critical Review over the Removal of Paracetamol (Acetaminophen) from Synthetic Waters and Real Wastewaters by Direct, Hybrid Catalytic, and Sequential Ozonation Processes. Chemosphere. 2023;313:137411. - PubMed
-
- Brillas E., Manuel Peralta-Hernández J. Removal of Paracetamol (Acetaminophen) by Photocatalysis and Photoelectrocatalysis. A Critical Review. Sep. Purif. Technol. 2023;309:122982.
-
- Al-howri B.M., Azha S.F., Shamsudin M.S., Hamid N.A., Alsobaai A.M., Ismail S. Paracetamol in Diverse Water Sources: Health Hazards and Treatment Efficacy Emphasizing Adsorption Techniques—A Review. Int. J. Environ. Sci. Technol. 2024;21:9743–9762. doi: 10.1007/s13762-024-05581-0. - DOI
-
- Ghanbari F., Hassani A., Wacławek S., Wang Z., Matyszczak G., Lin K.Y.A., Dolatabadi M. Insights into Paracetamol Degradation in Aqueous Solutions by Ultrasound-Assisted Heterogeneous Electro-Fenton Process: Key Operating Parameters, Mineralization and Toxicity Assessment. Sep. Purif. Technol. 2021;266:118533. doi: 10.1016/j.seppur.2021.118533. - DOI
-
- Eniola J.O., Kumar R., Barakat M.A., Rashid J. A Review on Conventional and Advanced Hybrid Technologies for Pharmaceutical Wastewater Treatment. J. Clean. Prod. 2022;356:131826.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

