Safety, efficacy, and cost-effectiveness evaluation of systemic treatments for refractory colorectal cancer: a systematic review and modeling study
- PMID: 40214895
- PMCID: PMC11987259
- DOI: 10.1186/s13561-025-00622-x
Safety, efficacy, and cost-effectiveness evaluation of systemic treatments for refractory colorectal cancer: a systematic review and modeling study
Abstract
Objectives: To conduct pooled estimates and comparative evaluations of safety and efficacy, alongside cost-effectiveness and value-based pricing analyses, for systemic treatments recommended by the National Comprehensive Cancer Network in refractory colorectal cancer.
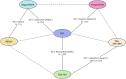
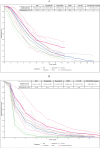
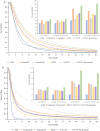
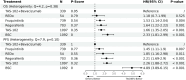
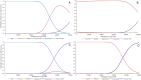
Methods: A comprehensive search for related randomized controlled trials was conducted on PubMed, EMBASE, the Cochrane Library, and ClinicalTrials.gov. Safety was evaluated by aggregating treatment-related adverse events (TRAEs) and performing Bayesian network meta-analysis (NMA) for indirect comparisons. Pooled survival estimates of overall survival (OS) and progression-free survival (PFS) were conducted to assess treatment efficacy. For NMA of OS and PFS, time-variant fractional polynomial models were employed as the primary analysis, with Cox proportional hazards models used for result validation. Economic evaluations were performed using partitioned survival models from the US public sector perspective. Clinical parameters were sourced from meta-analyses; cost parameters included drug treatment, follow-up and administration, end-of-life care, and adverse event management expenses, which were obtained from the Federal Supply Schedule, public databases or published literature. Utility values were sourced from the CORRECT trial. Price simulations were also conducted. Robustness of results was confirmed by sensitivity and scenario analyses RESULTS: We included nine studies comprising 3,978 patients and incorporating six treatments recommended by NCCN, including best supportive care (BSC), regorafenib, regorafenib dose optimization (REDo), trifluridine/tipiracil (TAS-102), TAS-102 with bevacizumab (TAS-BEV), and fruquintinib. Targeted treatments increased serious TRAEs and grade 3 + TRAEs compared to BSC. However, no significant safety differences were found among the targeted therapies. Regarding efficacy, REDo led in median OS, while fruquintinib led in median PFS. NMA indicated that TAS-BEV had the greatest PFS and OS survival benefit, followed by fruquintinib and REDo. Cost-effectiveness analysis favored BSC as the least expensive and the most cost-effective profile. TAS-BEV had the greatest effectiveness, with TAS-102 being the most cost-effective among targeted therapies. For cost-effectiveness against BSC, the price reductions of TAS-102, fruquintinib, REDoS, regorafenib, and TAS-BEV were 39%, 24%, 14%, 8%, and 7%, respectively.
Conclusions: Targeted therapies have comparable safety; TAS-BEV is highly effective, TAS-102 is the top cost-effective targeted therapy. Treatment choice should balance individual patient needs with safety, efficacy, and cost.
Keywords: Cost-effectiveness; Efficacy; Individual patient data meta-analysis; Refractory colorectal cancer; Safety.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethical approval: This study does not involve human participants or animal subjects. Patient consent for publication: Not required. Provenance and peer review: Not commissioned; externally peer reviewed. Patient and public involvement: No patient involved. Permission to reproduce material from other sources: NA. Competing interests: The authors declare no competing interests.
Figures



Similar articles
-
Cost-effectiveness analysis of regorafenib dose optimization for refractory metastatic colorectal cancer.J Med Econ. 2025 Dec;28(1):655-663. doi: 10.1080/13696998.2025.2496068. Epub 2025 Apr 28. J Med Econ. 2025. PMID: 40265856
-
Comparison of the efficacy and safety of third-line treatments for metastatic colorectal cancer: a systematic review and network meta-analysis.Front Oncol. 2023 Sep 21;13:1269203. doi: 10.3389/fonc.2023.1269203. eCollection 2023. Front Oncol. 2023. PMID: 37810981 Free PMC article.
-
Value-Based Analysis of Therapies in Refractory Metastatic Colorectal Cancer in US.Clin Colorectal Cancer. 2022 Dec;21(4):277-284. doi: 10.1016/j.clcc.2022.09.003. Epub 2022 Sep 17. Clin Colorectal Cancer. 2022. PMID: 36216759
-
Efficacy and safety of trifluridine/tipiracil plus bevacizumab versus trifluridine/tipiracil monotherapy for refractory metastatic colorectal cancer: a retrospective cohort study.J Gastrointest Oncol. 2024 Apr 30;15(2):612-629. doi: 10.21037/jgo-24-98. Epub 2024 Apr 28. J Gastrointest Oncol. 2024. PMID: 38756644 Free PMC article.
-
Regorafenib, TAS-102, or fruquintinib for metastatic colorectal cancer: any difference in randomized trials?Int J Colorectal Dis. 2020 Feb;35(2):295-306. doi: 10.1007/s00384-019-03477-x. Epub 2019 Dec 17. Int J Colorectal Dis. 2020. PMID: 31848739
References
-
- Sung H, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. - PubMed
-
- Siegel RL, et al. Colorectal cancer statistics, 2023. CA Cancer J Clin. 2023;73(3):233–54. - PubMed
-
- Dekker E, et al. Colorectal cancer. Lancet. 2019;394(10207):1467–80. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

