Maternal Metabolic Health and Mother and Baby Health Outcomes (MAMBO): Protocol of a Prospective Observational Study
- PMID: 40215105
- PMCID: PMC12032496
- DOI: 10.2196/72542
Maternal Metabolic Health and Mother and Baby Health Outcomes (MAMBO): Protocol of a Prospective Observational Study
Abstract
Background: Metabolic disease is increasingly impacting women of reproductive age. In pregnancy, uncontrolled metabolic disease can result in offspring with major congenital anomalies, preterm birth, and abnormal fetal growth. Pregnancy also accelerates the complications of metabolic diseases in mothers resulting in an increased risk of premature cardiovascular events. Despite the convincing evidence that preconception care can largely mitigate the risks of metabolic disease in pregnancy, there are few data about how to identify the highest-risk women so that they can be connected with appropriate preconception care services.
Objective: The aim of the study is to determine the maternal phenotype that represents the highest risk of having adverse neonatal and maternal pregnancy outcomes.
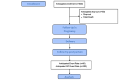
Methods: This will be a prospective cohort study of 500 women recruited in early pregnancy. The primary outcome is a composite of offspring born small for gestational age (SGA) or large for gestational age (LGA) (customized birthweight ≤10th and ≥90th centile for gestational age). Secondary outcomes are (1) composite of adverse neonatal birth outcomes (SGA, LGA, major congenital abnormalities, preterm birth [<37 weeks' gestation]) and (2) composite of new maternal metabolic outcomes (gestational diabetes, diabetes in pregnancy, type 2 diabetes [T2D] or prediabetes; gestational hypertension, preeclampsia, eclampsia or new essential hypertension after pregnancy; and gestational weight gain ≥20kg or new overweight/obesity at the 12-18 months postpartum visit). A multivariable logistic regression analysis will be conducted to identify candidate predictors of poor pregnancy outcomes due to metabolic disease. From this model, model coefficients and the associated 95% CIs will be extracted to derive the risk score for predicting the delivery of LGA/SGA offspring (primary outcome) and composites of adverse neonatal outcomes and maternal outcomes (secondary outcomes).
Results: Seed funding for the project was acquired in November 2022 and subsequent funding was acquired in May 2024. The first participant was recruited on March 23, 2023. At the time of manuscript submission, 402 participants have been recruited. Data analysis has not yet been performed. Results are expected to be published in the first half of 2027.
Conclusions: This is a prospective observational cohort study that intends to identify the metabolic disease risk factors, or combination of factors, that are most likely to cause adverse maternal and fetal health outcomes. These characteristics will be used to develop a risk calculator which will assist in identifying the highest risk women and in triaging them to appropriate services. The study has been approved by the institutional Human Research Ethics Committee (HREC/90080/MH-2022).
Trial registration: Australian New Zealand Clinical Trials Registry ACTRN12623000037606; https://tinyurl.com/yeytsxtp.
International registered report identifier (irrid): DERR1-10.2196/72542.
Keywords: diabetes; hypertension; large for gestational age; metabolic disease; obesity; preconception; pregnancy outcomes; small for gestational age.
©Sarah A L Price, Digsu N Koye, Alice Lewin, Alison Nankervis, Stefan C Kane. Originally published in JMIR Research Protocols (https://www.researchprotocols.org), 11.04.2025.
Conflict of interest statement
Conflicts of Interest: None declared.
Figures
Similar articles
-
Development and validation of prediction models for fetal growth restriction and birthweight: an individual participant data meta-analysis.Health Technol Assess. 2024 Aug;28(47):1-119. doi: 10.3310/DABW4814. Health Technol Assess. 2024. PMID: 39252507 Free PMC article.
-
Planned delivery for pre-eclampsia between 34 and 37 weeks of gestation: the PHOENIX RCT.Health Technol Assess. 2023 Dec;27(28):1-22. doi: 10.3310/CWWH0622. Health Technol Assess. 2023. PMID: 36547875 Free PMC article. Clinical Trial.
-
Association of maternal polycystic ovary syndrome and diabetes with preterm birth and offspring birth size: a population-based cohort study.Hum Reprod. 2022 May 30;37(6):1311-1323. doi: 10.1093/humrep/deac050. Hum Reprod. 2022. PMID: 35348682 Free PMC article.
-
Gestational weight gain during the second and third trimesters and adverse pregnancy outcomes, results from a prospective pregnancy cohort in urban Tanzania.Reprod Health. 2022 Jun 16;19(1):140. doi: 10.1186/s12978-022-01441-7. Reprod Health. 2022. PMID: 35710384 Free PMC article. Clinical Trial.
-
Omega-3 fatty acid addition during pregnancy.Cochrane Database Syst Rev. 2018 Nov 15;11(11):CD003402. doi: 10.1002/14651858.CD003402.pub3. Cochrane Database Syst Rev. 2018. PMID: 30480773 Free PMC article.
References
-
- Australian Government. Sydney, Australia: Australian Institute of Health and Welfare; 2024. [2025-03-21]. Australia's mothers and babies. https://www.aihw.gov.au/reports-data/population-groups/mothers-babies/ov... .
-
- CiteSeer. Australian Institute of Health and Welfare; 2023. [2025-03-14]. Australia's mothers and babies. https://citeseerx.ist.psu.edu/document?repid=rep1&type=pdf&doi=407ab9f2d... .
MeSH terms
LinkOut - more resources
Full Text Sources
Medical