Association of eosinophil-to-monocyte ratio with asthma exacerbations in adults: a cross-sectional analysis of NHANES data
- PMID: 40217197
- PMCID: PMC11992703
- DOI: 10.1186/s12890-025-03617-w
Association of eosinophil-to-monocyte ratio with asthma exacerbations in adults: a cross-sectional analysis of NHANES data
Abstract
Background: The eosinophil-to-monocyte ratio (EMR) has emerged as a promising biomarker for assessing inflammation in various diseases, and this study aims to investigate its potential in predicting asthma exacerbations.
Methods: This cross-sectional study used data from the National Health and Nutrition Examination Survey (NHANES) 1999-2020. A total of 4,738 adults were included in the analysis, and weighted analyses were performed to ensure a representative sample of the general population. The relationship between EMR and asthma exacerbation risk was assessed using multivariable logistic regression with progressively adjusted covariates across multiple models. Subgroup analyses were performed by stratifying key covariates to explore interactions. Restricted cubic spline (RCS) analysis was applied to evaluate non-linear relationships. Sensitivity analyses confirmed the robustness and reliability of the results.
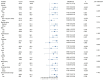
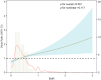
Results: Elevated EMR levels were significantly associated with an increased risk of asthma exacerbations (p < 0.001 in all models). In the highest EMR quartile (Q4), the odds ratio for exacerbation compared to the lowest quartile (Q1) was 1.54 (95% CI: 1.23, 1.93) in Model 1, increasing to 1.56 (95% CI: 1.24, 1.97) in Model 2 and 1.58 (95% CI: 1.24, 2.02) in Model 3, after further adjustments. Subgroup analyses showed consistent associations across various characteristics (all p for interaction > 0.05), while RCS analysis revealed a linear relationship without threshold effects (p for nonlinear > 0.05).
Conclusion: EMR demonstrates strong potential as a biomarker for predicting asthma exacerbations, with implications for personalized asthma management.
Keywords: Asthma; Biomarker; Eosinophil-to-monocyte ratio (EMR); Exacerbation; NHANES.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: NHANES protocols and consent procedures were approved by the NCHS Research Ethics Review Board. Written informed consent was obtained from all participants, and the study adhered to STROBE guidelines to maintain rigorous methodological and ethical standards. For further details, please refer to NCHS NHANES and NCHS ERB Approval. Consent for publication: No individual personal information is disclosed in this publication, and all data have been de-identified to maintain participant confidentiality. Competing interests: The authors declare no competing interests. Clinical trial number: Not applicable.
Figures
References
-
- Busse WW, Lemanske RF Jr., Asthma. N Engl J Med. 2001;344(5):350–62. 10.1056/NEJM200102013440507. - PubMed
-
- Bousquet J, et al. Asthma. From bronchoconstriction to airways inflammation and remodeling. Am J Respir Crit Care Med. 2000;161(5):1720–45. 10.1164/ajrccm.161.5.9903102. - PubMed
-
- Pavord ID, et al. Mepolizumab for severe eosinophilic asthma (DREAM): a multicentre, double-blind, placebo-controlled trial. Lancet. 2012;380(9842):651–9. 10.1016/S0140-6736(12)60988-X. - PubMed
-
- Bel EH, et al. Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N Engl J Med. 2014;371(13):1189–97. 10.1056/NEJMoa1403291. Epub 2014 Sep 8. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

