Research progress of connexins in epileptogensis
- PMID: 40217413
- PMCID: PMC11960343
- DOI: 10.1186/s42494-025-00203-9
Research progress of connexins in epileptogensis
Abstract
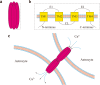
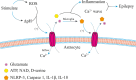
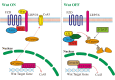
Epilepsy, a chronic neurological disorder, is characterized by dysfunction in neural networks. Gap junctions and hemichannels, which are integral to the astrocyte connection network, play a critical role in epilepsy. Connexins, the components of astrocyte gap junctions and hemichannels, can be activated to transfer glutamate, adenosine triphosphate, and other chemicals, potentially leading to seizures. Connexins therefore hold significant potential for epilepsy treatment. This review focuses on connexin 43 and provides a brief overview of other connexins and pannexin 1. Understanding the relationship between connexins and epilepsy offers theoretical support for developing new antiseizure medications.
Keywords: Connexin; Epilepsy; Gap junction; Hemichannel; Wnt pathway.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: All authors declare that there are no conflicts of interest.
Figures

Similar articles
-
Astroglial connexins in epileptogenesis.Seizure. 2021 Jan;84:122-128. doi: 10.1016/j.seizure.2020.11.022. Epub 2020 Dec 8. Seizure. 2021. PMID: 33348235 Review.
-
Connexins and pannexins: At the junction of neuro-glial homeostasis & disease.J Neurosci Res. 2018 Jan;96(1):31-44. doi: 10.1002/jnr.24088. Epub 2017 Jun 5. J Neurosci Res. 2018. PMID: 28580666 Free PMC article. Review.
-
Connexin and pannexin hemichannels in inflammatory responses of glia and neurons.Brain Res. 2012 Dec 3;1487:3-15. doi: 10.1016/j.brainres.2012.08.042. Epub 2012 Sep 10. Brain Res. 2012. PMID: 22975435 Free PMC article. Review.
-
Antibodies targeting extracellular domain of connexins for studies of hemichannels.Neuropharmacology. 2013 Dec;75:525-32. doi: 10.1016/j.neuropharm.2013.02.021. Epub 2013 Mar 13. Neuropharmacology. 2013. PMID: 23499293 Free PMC article. Review.
-
Connexin hemichannels and pannexin channels in toxicity: Recent advances and mechanistic insights.Toxicology. 2023 Apr;488:153488. doi: 10.1016/j.tox.2023.153488. Epub 2023 Mar 12. Toxicology. 2023. PMID: 36918108 Review.
References
-
- Thijs RD, Surges R, O’Brien TJ, Sander JW. Epilepsy in adults. Lancet. 2019;393(10172):689–701. - PubMed
-
- Walker MC, Kullmann DM. Optogenetic and chemogenetic therapies for epilepsy. Neuropharmacology. 2020;168:107751. - PubMed
-
- Kalilani L, Sun X, Pelgrims B, Noack-Rink M, Villanueva V. The epidemiology of drug-resistant epilepsy: a systematic review and meta-analysis. Epilepsia. 2018;59(12):2179–93. - PubMed
-
- Mesraoua B, Brigo F, Lattanzi S, Abou-Khalil B, Al Hail H, Asadi-Pooya AA. Drug-resistant epilepsy: definition, pathophysiology, and management. J Neurol Sci. 2023;452:120766. - PubMed
-
- Wang S, Zhao M, Li T, Zhang C, Zhou J, Wang M, et al. Long-term efficacy and cognitive effects of bilateral hippocampal deep brain stimulation in patients with drug-resistant temporal lobe epilepsy. Neurol Sci. 2021;42(1):225–33. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
