A novel approach utilizing spirocyclic thiopyrimidinone compounds against herpes simplex virus with underlying antiviral mechanisms of action
- PMID: 40217544
- PMCID: PMC11987438
- DOI: 10.1186/s12985-025-02707-9
A novel approach utilizing spirocyclic thiopyrimidinone compounds against herpes simplex virus with underlying antiviral mechanisms of action
Abstract
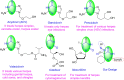
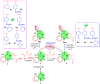
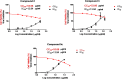
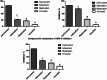
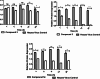
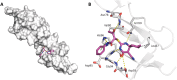
In recent decades, viral outbreaks have significantly threatened global health, with herpes simplex virus type 2 (HSV-2) being one of the most prevalent infections. This study evaluated novel spiropyrimidine derivatives as potential antiviral agents against HSV-2, building on previous research that examined spirocyclic thiopyrimidinone derivatives against human coronavirus 229E (hCoV-229E). Among the eleven synthesized compounds, spiropyrimidinone derivative 3 demonstrated promising antiviral activity, with a selectivity index of 11.2. The drug mechanism of infection studies indicated that compound 3 primarily inhibits HSV-2 at the viral adsorption stage, achieving approximately 83% inhibition and reducing viral multiplication by 34%. Its efficacy is linked to its diketone moiety, which is known for its ability to enhance antiviral effects. Furthermore, the effect of compound 3 on viral inhibition is reflected in the level of caspase-3 protein expression, revealing that the apoptotic pathway is modulated. Docking studies revealed multiple interactions with herpes virus entry mediator (HVEM), indicating its potential as an entry inhibitor. These findings confirm that compound 3 could be a potential candidate for further development in HSV-2 antiviral therapy.
Keywords: Antiviral activity; Herpes simplex virus; Mechanism of action; Spirocyclic thiopyrimidinones.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: We hereby grant our consent for the publication of our manuscript and confirm that all coauthors have agreed to this submission. Competing interests: The authors declare no competing interests.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

