High-Fat Diet in Perinatal Period Promotes Liver Steatosis and Low Desaturation Capacity of Polyunsaturated Fatty Acids in Dams: A Link with Anxiety-Like Behavior in Rats
- PMID: 40218938
- PMCID: PMC11990584
- DOI: 10.3390/nu17071180
High-Fat Diet in Perinatal Period Promotes Liver Steatosis and Low Desaturation Capacity of Polyunsaturated Fatty Acids in Dams: A Link with Anxiety-Like Behavior in Rats
Abstract
Background/objectives: This study investigates the effects of a high-fat diet (HFD) during pregnancy and lactation on maternal and offspring health, focusing on behavioral, metabolic, and fatty acid composition outcomes in a rat model.
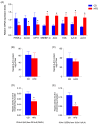
Methods: Twelve female Sprague-Dawley rats were fed either a control diet, CD (n = 6), or HFD (n = 6) for 12 weeks, encompassing mating, gestation, and lactation periods (18 weeks). Anxiety-like behavior, maternal behavior, depression-like behavior, and social play were studied. Post mortem, the liver function, hepatic steatosis, and fatty acid composition (erythrocytes, liver, adipose tissue) were evaluated. In regard to desaturase enzymes (Δ-6D and Δ-5D), liver activity, protein mass, and gene expression (RT-PCR) were analyzed. Additionally, gene expression of PPAR-α, ACOX, CPT1-α, SREBP-1c, ACC, and FAS was assessed. Statistical analysis was performed using Student's t-test, mean ± SD (p < 0.05).
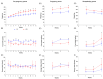
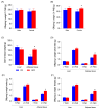
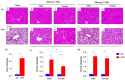
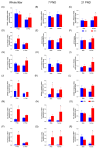
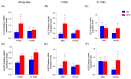
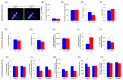
Results: The HFD significantly increased maternal weight and anxiety-like behavior while reducing social interactions exclusively in male offspring (p < 0.05). It also led to a significant decrease in the synthesis and content of n-3 PUFAs in the analyzed tissues, induced hepatic steatosis, and upregulated the expression of pro-lipogenic genes in the maternal liver.
Conclusions: These findings suggest that long-term HFD consumption alters tissue fatty acid composition, disrupts metabolic homeostasis, and contributes to behavioral changes, increasing anxiety-like behaviors in pregnant dams and reducing social interactions in male offspring. Overall, this study provides further insight into the detrimental effects of HFD consumption during the perinatal period.
Keywords: alpha-linolenic acid; anxiety; docosahexaenoic acid; fatty acid metabolism; high-fat diet; linoleic acid; n-3 polyunsaturated fatty acid; pregnancy.
Conflict of interest statement
All authors declare that they have no conflicts of interest.
Figures

Similar articles
-
Role of maternal tissue in the synthesis of polyunsaturated fatty acids in response to a lipid-deficient diet during pregnancy and lactation in rats.Gene. 2014 Oct 1;549(1):7-23. doi: 10.1016/j.gene.2014.06.057. Epub 2014 Jul 19. Gene. 2014. PMID: 25046614
-
Hydroxytyrosol prevents reduction in liver activity of Δ-5 and Δ-6 desaturases, oxidative stress, and depletion in long chain polyunsaturated fatty acid content in different tissues of high-fat diet fed mice.Lipids Health Dis. 2017 Apr 11;16(1):64. doi: 10.1186/s12944-017-0450-5. Lipids Health Dis. 2017. PMID: 28395666 Free PMC article.
-
Reduction in the desaturation capacity of the liver in mice subjected to high fat diet: Relation to LCPUFA depletion in liver and extrahepatic tissues.Prostaglandins Leukot Essent Fatty Acids. 2015 Jul;98:7-14. doi: 10.1016/j.plefa.2015.04.002. Epub 2015 Apr 13. Prostaglandins Leukot Essent Fatty Acids. 2015. PMID: 25910408
-
Maternal conjugated linoleic acid consumption prevented TAG alterations induced by a high-fat diet in male adult rat offspring.Br J Nutr. 2020 Aug 14;124(3):286-295. doi: 10.1017/S0007114520001166. Epub 2020 Apr 1. Br J Nutr. 2020. PMID: 32234086
-
Maternal exposure to high-fat diet modifies anxiety-like/depression-like behaviors and compounds of Serotonergic System in offspring: A preclinical systematic review.Int J Dev Neurosci. 2021 Aug;81(5):371-385. doi: 10.1002/jdn.10110. Epub 2021 May 2. Int J Dev Neurosci. 2021. PMID: 33788300
References
-
- Abdelrahman M.A., Osama H., Saeed H., Madney Y.M., Harb H.S., Abdelrahim M.E.A. Impact of n-3 polyunsaturated fatty acid intake in pregnancy on maternal health and birth outcomes: Systematic review and meta-analysis from randomized controlled trails. Arch. Gynecol. Obstet. 2022;307:249–262. - PubMed
-
- Ortiz M., Sánchez F., Álvarez D., Flores C., Salas-Pérez F., Valenzuela R., Cantin C., Leiva A., Crisosto N., Maliqueo M. Association between maternal obesity, essential fatty acids and biomarkers of fetal liver function. Prostaglandins Leukot. Essent. Fatty Acids. 2023;190:102541. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

