Fibromodulin selectively accelerates myofibroblast apoptosis in cutaneous wounds by enhancing interleukin 1β signaling
- PMID: 40221432
- PMCID: PMC11993684
- DOI: 10.1038/s41467-025-58906-z
Fibromodulin selectively accelerates myofibroblast apoptosis in cutaneous wounds by enhancing interleukin 1β signaling
Abstract
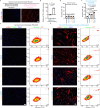
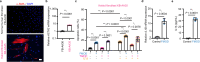
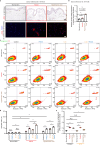
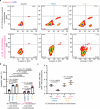
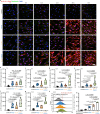
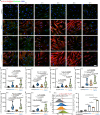
Activated myofibroblasts deposit extracellular matrix material to facilitate rapid wound closure that can heal scarlessly during fetal development. However, adult myofibroblasts exhibit a relatively long life and persistent function, resulting in scarring. Thus, understanding how fetal and adult tissue regeneration differs may serve to identify factors that promote more optimal wound healing in adults with little or less scarring. We previously found that matricellular proteoglycan fibromodulin is one such factor promoting more optimal repair, but the underlying molecular and cellular mechanisms for these effects have not been fully elucidated. Here, we find that fibromodulin induces myofibroblast apoptosis after wound closure to reduce scarring in small and large animal models. Mechanistically, fibromodulin accelerates and prolongs the formation of the interleukin 1β-interleukin 1 receptor type 1-interleukin 1 receptor accessory protein ternary complex to increase the apoptosis of myofibroblasts and keloid- and hypertrophic scar-derived cells. As the persistence of myofibroblasts during tissue regeneration is a key cause of fibrosis in most organs, fibromodulin represents a promising, broad-spectrum anti-fibrotic therapeutic.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: Drs. Kang Ting, Chia Soo, and Zhong Zheng are inventors of fibromodulin-related patents assigned to UCLA. Drs. Kang Ting, Chia Soo, and Zhong Zheng are founders of Scarless Laboratories Inc. and Saint Therapeutics Inc., which sublicense fibromodulin-related patents from the UC Regents, who also hold equity in the company. Drs. Kang Ting, Chia Soo, and Zhong Zheng are also officers of Scarless Laboratories, Inc. and Saint Therapeutics Inc. The remaining authors declare no competing interests.
Figures




References
-
- Cass, D. L., Sylvester, K. G., Yang, E. Y., Crombleholme, T. M. & Adzick, N. S. Myofibroblast persistence in fetal sheep wounds is associated with scar formation. J. Pediatr. Surg.32, 1017–1021 (1997). discussion 1021-1012. - PubMed
-
- Hinz, B. The myofibroblast: paradigm for a mechanically active cell. J. Biomech.43, 146–155 (2010). - PubMed
-
- Sandulache, V. C., Parekh, A., Dohar, J. E. & Hebda, P. A. Fetal dermal fibroblasts retain a hyperactive migratory and contractile phenotype under 2-and 3-dimensional constraints compared to normal adult fibroblasts. Tissue Eng.13, 2791–2801 (2007). - PubMed
MeSH terms
Substances
Grants and funding
- SB1DE026972/U.S. Department of Health & Human Services | NIH | National Institute of Dental and Craniofacial Research (NIDCR)
- SB1 DE026972/DE/NIDCR NIH HHS/United States
- R44DE024692/U.S. Department of Health & Human Services | NIH | National Institute of Dental and Craniofacial Research (NIDCR)
- R44AR064126/U.S. Department of Health & Human Services | NIH | National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS)
- R44 AR064126/AR/NIAMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

