Redox signaling modulates axonal microtubule organization and induces a specific phosphorylation signature of microtubule-regulating proteins
- PMID: 40222271
- PMCID: PMC12019850
- DOI: 10.1016/j.redox.2025.103626
Redox signaling modulates axonal microtubule organization and induces a specific phosphorylation signature of microtubule-regulating proteins
Abstract
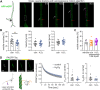
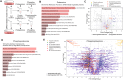
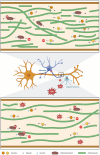
Many life processes are regulated by physiological redox signaling, but excessive oxidative stress can damage biomolecules and contribute to disease. Neuronal microtubules are critically involved in axon homeostasis, regulation of axonal transport, and neurodegenerative processes. However, whether and how physiological redox signaling affects axonal microtubules is largely unknown. Using live cell imaging and super-resolution microscopy, we show that subtoxic concentrations of the central redox metabolite hydrogen peroxide increase axonal microtubule dynamics, alter the structure of the axonal microtubule array, and affect the efficiency of axonal transport. We report that the mitochondria-targeting antioxidant SkQ1 and the microtubule stabilizer EpoD abolish the increase in microtubule dynamics. We found that hydrogen peroxide specifically modulates the phosphorylation state of microtubule-regulating proteins, which differs from arsenite as an alternative stress inducer, and induces a largely non-overlapping phosphorylation pattern of MAP1B as a main target. Cell-wide phosphoproteome analysis revealed signaling pathways that are inversely activated by hydrogen peroxide and arsenite. In particular, hydrogen peroxide treatment was associated with kinases that suppress apoptosis and regulate brain metabolism (PRKDC, CK2, PDKs), suggesting that these pathways play a central role in physiological redox signaling and modulation of axonal microtubule organization. The results suggest that the redox metabolite and second messenger hydrogen peroxide induces rapid and local reorganization of the microtubule array in response to mitochondrial activity or as a messenger from neighboring cells by activating specific signaling cascades.
Keywords: Axon; Hydrogen peroxide; Microtubules; Redox signalling; Tau.
Copyright © 2025 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare no competing interests.
Figures




References
-
- Penazzi L., Bakota L., Brandt R. Microtubule dynamics in neuronal development, plasticity, and neurodegeneration. Int. Rev. Cell. Mol. Biol. 2016;321:89–169. - PubMed
-
- Uhlen M., Fagerberg L., Hallstrom B.M., Lindskog C., Oksvold P., Mardinoglu A., Sivertsson A., Kampf C., Sjostedt E., Asplund A., Olsson I., Edlund K., Lundberg E., Navani S., Szigyarto C.A., Odeberg J., Djureinovic D., Takanen J.O., Hober S., Alm T., Edqvist P.H., Berling H., Tegel H., Mulder J., Rockberg J., Nilsson P., Schwenk J.M., Hamsten M., von Feilitzen K., Forsberg M., Persson L., Johansson F., Zwahlen M., von Heijne G., Nielsen J., Ponten F. Proteomics. Tissue-based map of the human proteome. Science. 2015;347 - PubMed
-
- Janke C., Magiera M.M. The tubulin code and its role in controlling microtubule properties and functions. Nat. Rev. Mol. Cell Biol. 2020;21:307–326. - PubMed
-
- Arendt T., Stieler J.T., Holzer M. Tau and tauopathies. Brain Res. Bull. 2016;126:238–292. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

