Emerging therapeutic strategies in glioblastsoma: drug repurposing, mechanisms of resistance, precision medicine, and technological innovations
- PMID: 40223032
- PMCID: PMC11994545
- DOI: 10.1007/s10238-025-01631-0
Emerging therapeutic strategies in glioblastsoma: drug repurposing, mechanisms of resistance, precision medicine, and technological innovations
Abstract
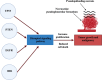
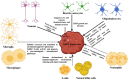
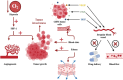
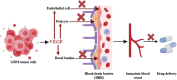
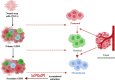
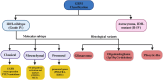
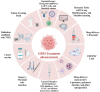
Glioblastoma (GBM) is an aggressive Grade IV brain tumor with a poor prognosis. It results from genetic mutations, epigenetic changes, and factors within the tumor microenvironment (TME). Traditional treatments like surgery, radiotherapy, and chemotherapy provide limited survival benefits due to the tumor's heterogeneity and resistance mechanisms. This review examines novel approaches for treating GBM, focusing on repurposing existing medications such as antipsychotics, antidepressants, and statins for their potential anti-GBM effects. Advances in molecular profiling, including next-generation sequencing, artificial intelligence (AI), and nanotechnology-based drug delivery, are transforming GBM diagnosis and treatment. The TME, particularly GBM stem cells and immune evasion, plays a key role in therapeutic resistance. Integrating multi-omics data and applying precision medicine show promise, especially in combination therapies and immunotherapies, to enhance clinical outcomes. Addressing challenges such as drug resistance, targeting GBM stem cells, and crossing the blood-brain barrier is essential for improving treatment efficacy. While current treatments offer limited benefits, emerging strategies such as immunotherapies, precision medicine, and drug repurposing show significant potential. Technologies like liquid biopsies, AI-powered diagnostics, and nanotechnology could help overcome obstacles like the blood-brain barrier and GBM stem cells. Ongoing research into combination therapies, targeted drug delivery, and personalized treatments is crucial. Collaborative efforts and robust clinical trials are necessary to translate these innovations into effective therapies, offering hope for improved survival and quality of life for GBM patients.
Keywords: Artificial intelligence; Blood–brain barrier; Nanotechnology; Next-generation sequencing; Precision medicine; Tumor microenvironment.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: The authors declare no competing interests. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable.
Figures




References
-
- Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, et al. The 2016 world health organization classification of tumors of the central nervous system: a summary. Acta Neuropathol. 2016;131(6):803–20. - PubMed
-
- McKinnon C, Nandhabalan M, Murray SA, Plaha P. Glioblastoma: clinical presentation, diagnosis, and management. BMJ. 2021;374:574012. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

