Global patterns of hemophilia drug trials, hemophilia care, and health care measures
- PMID: 40224274
- PMCID: PMC11992419
- DOI: 10.1016/j.rpth.2025.102714
Global patterns of hemophilia drug trials, hemophilia care, and health care measures
Abstract
Background: Drug trials are vital to establish safe and effective treatments for congenital hemophilia, a bleeding disorder that affects about 800,000 males worldwide. The global distribution of hemophilia drug trials (HDTs) and their alignment with hemophilia care is unknown.
Objectives: This study aimed to evaluate the global distribution of HDTs and its association with hemophilia care.
Methods: In this cross-sectional study, HDTs conducted between 2007 and 2022 were selected from the clinicaltrials.gov database. The density of trials per 1000 expected males with hemophilia (eMwH) was assessed according to hemophilia care measures (factor VIII and IX utilization per 1000 eMwH) derived from World Federation of Hemophilia data.
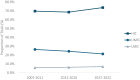
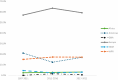
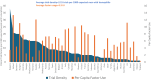
Results: Among 124 trials, 55 countries were represented, with an average of 7.9 countries per trial. Most HDT sites were in high-income (74.4%) or upper middle (20.1%)-income countries. The number of sites in lower-middle-income countries doubled, from 12 in 2007-2011 to 30 in 2017-2022-a nonsignificant increase from 5.8% to 7.0% (P = .53). Factor utilization was substantially reduced in lower-middle (0.4 international units [IUs] per 1000 eMwH) and upper middle (2.8 IUs per 1000 eMwH) compared with high (6.8 IUs per 1000 eMwH) income countries. HDT density was moderately correlated with factor usage (r = 0.436; P ≤ .001).
Conclusion: Most HDT sites were located in high-income countries, although a substantial proportion were in upper middle-income countries. A small but increasing number of trials were conducted in lower-middle-income countries, where factor usage is relatively low. This study provides evidence on the global distribution of HDT and raises questions regarding the generalizability, barriers, opportunities, and ethics of trials for a rare bleeding disorder.
Keywords: clinical trials; global health; health equity; hemophilia.
© 2025 The Authors.
Figures

Similar articles
-
The World Federation of Hemophilia World Bleeding Disorders Registry: insights from the first 10,000 patients.Res Pract Thromb Haemost. 2023 Nov 20;7(8):102264. doi: 10.1016/j.rpth.2023.102264. eCollection 2023 Nov. Res Pract Thromb Haemost. 2023. PMID: 38193052 Free PMC article.
-
A study of reported factor IX use around the world.Haemophilia. 2011 May;17(3):446-55. doi: 10.1111/j.1365-2516.2010.02461.x. Epub 2011 Feb 7. Haemophilia. 2011. PMID: 21299742
-
Prevalence, incidence, and factor concentrate usage trends of hemophiliacs in Taiwan.Yonsei Med J. 2013 Jan 1;54(1):71-80. doi: 10.3349/ymj.2013.54.1.71. Yonsei Med J. 2013. PMID: 23225801 Free PMC article.
-
Global prophylaxis trends in hemophilia: a macroeconomic analysis and its association with world development indicators.Expert Rev Hematol. 2024 Dec;17(12):947-956. doi: 10.1080/17474086.2024.2429606. Epub 2024 Nov 18. Expert Rev Hematol. 2024. PMID: 39535178 Review.
-
Achieving access to haemophilia care in low-income and lower-middle-income countries: expanded Humanitarian Aid Program of the World Federation of Hemophilia after 5 years.Lancet Haematol. 2022 Sep;9(9):e689-e697. doi: 10.1016/S2352-3026(22)00209-5. Lancet Haematol. 2022. PMID: 36055333 Review.
References
-
- Croteau S.E., Wang M., Wheeler A.P. 2021 clinical trials update: Innovations in hemophilia therapy. Am J Hematol. 2021;96:128–144. - PubMed
-
- World Federation of Hemophilia. Achieving global impact: strategic plan 2021-2025. World Federation of Hemophilia.
-
- World Federation of Hemophilia . World Federation of Hemophilia; October 2022. World Federation of Hemophilia report on the annual global survey 2021.
LinkOut - more resources
Full Text Sources

