Immune response recalibration using immune therapy and biomimetic nano-therapy against high-grade gliomas and brain metastases
- PMID: 40224727
- PMCID: PMC11987628
- DOI: 10.1016/j.ajps.2025.101021
Immune response recalibration using immune therapy and biomimetic nano-therapy against high-grade gliomas and brain metastases
Abstract
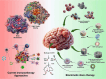
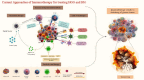
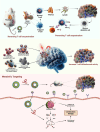
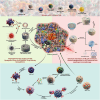
Although with aggressive standards of care like surgical resection, chemotherapy, and radiation, high-grade gliomas (HGGs) and brain metastases (BM) treatment has remained challenging for more than two decades. However, technological advances in this field and immunotherapeutic strategies have revolutionized the treatment of HGGs and BM. Immunotherapies like immune checkpoint inhibitors, CAR-T targeting, oncolytic virus-based therapy, bispecific antibody treatment, and vaccination approaches, etc., are emerging as promising avenues offering new hope in refining patient's survival benefits. However, selective trafficking across the blood-brain barrier (BBB), immunosuppressive tumor microenvironment (TME), metabolic alteration, and tumor heterogeneity limit the therapeutic efficacy of immunotherapy for HGGs and BM. Furthermore, to address this concern, the NanoBioTechnology-based bioinspired delivery system has been gaining tremendous attention in recent years. With technological advances such as Trojan horse targeting and infusing/camouflaging nanoparticles surface with biological molecules/cells like immunocytes, erythrocytes, platelets, glioma cell lysate and/or integrating these strategies to get hybrid membrane for homotypic recognition. These biomimetic nanotherapy offers advantages over conventional nanoparticles, focusing on greater target specificity, increased circulation stability, higher active loading capacity, BBB permeability (inherent inflammatory chemotaxis of neutrophils), decreased immunogenicity, efficient metabolism-based combinatorial effects, and prevention of tumor recurrence by induction of immunological memory, etc. provide new age of improved immunotherapies outcomes against HGGs and BM. In this review, we emphasize on neuro-immunotherapy and the versatility of these biomimetic nano-delivery strategies for precise targeting of hard-to-treat and most lethal HGGs and BM. Moreover, the challenges impeding the clinical translatability of these approaches were addressed to unmet medical needs of brain cancers.
Keywords: Biomimetic nanoparticles; Blood-brain-barrier; Brain metastases; Clinical translatability; High-grade glioma; Immunotherapy.
© 2025 Shenyang Pharmaceutical University. Published by Elsevier B.V.
Conflict of interest statement
All the authors declared that the investigation was conducted in the absence of any financial/commercial interest that could be constructed as a potential conflict.
Figures




Similar articles
-
Combinatorial approaches to effective therapy in glioblastoma (GBM): Current status and what the future holds.Int Rev Immunol. 2022;41(6):582-605. doi: 10.1080/08830185.2022.2101647. Epub 2022 Aug 8. Int Rev Immunol. 2022. PMID: 35938932 Review.
-
Immunotherapy: a promising approach for glioma treatment.Front Immunol. 2023 Sep 7;14:1255611. doi: 10.3389/fimmu.2023.1255611. eCollection 2023. Front Immunol. 2023. PMID: 37744349 Free PMC article. Review.
-
CAR-T Therapy for Pediatric High-Grade Gliomas: Peculiarities, Current Investigations and Future Strategies.Front Immunol. 2022 May 4;13:867154. doi: 10.3389/fimmu.2022.867154. eCollection 2022. Front Immunol. 2022. PMID: 35603195 Free PMC article. Review.
-
Overcoming immunotherapy resistance in glioblastoma: challenges and emerging strategies.Front Pharmacol. 2025 Mar 28;16:1584688. doi: 10.3389/fphar.2025.1584688. eCollection 2025. Front Pharmacol. 2025. PMID: 40223940 Free PMC article. Review.
-
Fabrication of Biomimetic Hybrid Liposomes via Microfluidic Technology: Homotypic Targeting and Antitumor Efficacy Studies in Glioma Cells.Int J Nanomedicine. 2024 Dec 8;19:13217-13233. doi: 10.2147/IJN.S489872. eCollection 2024. Int J Nanomedicine. 2024. PMID: 39679250 Free PMC article.
References
-
- Widodo S.S., Dinevska M., Cuzcano L., Papanicolaou M., Cox T.R., Stylli S.S., et al. Spatial analysis of the metastatic brain tumor immune and extracellular matrix microenvironment. Adv Cancer Biol - Metastasis. 2023;7
-
- Vishnoi M., Scranton R.A., Asante S.K., Rostomily R.C. In: Central nervous system metastases. Ramakrishna R, Magge R, Baaj A, Knisely J, editors. Springer International Publishing; Cham: 2020. Basic biology of brain metastasis; pp. 19–35.
Publication types
LinkOut - more resources
Full Text Sources

