miRNA-based regulation in growth plate cartilage: mechanisms, targets, and therapeutic potential
- PMID: 40225327
- PMCID: PMC11985438
- DOI: 10.3389/fendo.2025.1530374
miRNA-based regulation in growth plate cartilage: mechanisms, targets, and therapeutic potential
Abstract
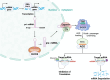
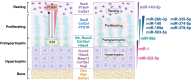
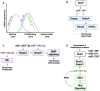
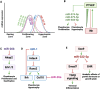
MicroRNAs (miRNAs) are critical regulators of the skeleton. In the growth plate, these small non-coding RNAs modulate gene networks that drive key stages of chondrogenesis, including proliferation, differentiation, extracellular matrix synthesis and hypertrophy. These processes are orchestrated through the interaction of pivotal pathways including parathyroid hormone-related protein (PTHrP), Indian hedgehog (IHH), and bone morphogenetic protein (BMP) signaling. This review highlights the miRNA-mRNA target networks essential for chondrocyte differentiation. Many miRNAs are differentially expressed in resting, proliferating and hypertrophic cartilage zones. Moreover, differential enrichment of specific miRNAs in matrix vesicles is also observed, providing means for chondrocytes to influence the function and differentiation of their neighbors by via matrix vesicle protein and RNA cargo. Notably, miR-1 and miR-140 emerge as critical modulators of chondrocyte proliferation and hypertrophy by regulating multiple signaling pathways, many of them downstream from their mutual target Hdac4. Demonstration that a human gain-of-function mutation in miR-140 causes skeletal dysplasia underscores the clinical relevance of understanding miRNA-mediated regulation. Further, miRNAs such as miR-26b have emerged as markers for skeletal disorders such as idiopathic short stature, showcasing the translational relevance of miRNAs in skeletal health. This review also highlights some miRNA-based therapeutic strategies, including innovative delivery systems that could target chondrocytes via cartilage affinity peptides, and potential applications related to treatment of physeal bony bridge formation in growing children. By synthesizing current research, this review offers a nuanced understanding of miRNA functions in growth plate biology and their broader implications for skeletal health. It underscores the translational potential of miRNA-based therapies in addressing skeletal disorders and aims to inspire further investigations in this rapidly evolving field.
Keywords: SOX9; chondrocyte; growth plate; matrix vesicles; miR-140; microRNA; regenerative medicine; skeletal dysplasia.
Copyright © 2025 Thakore and Delany.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
The G protein-coupled receptor ADGRG6 maintains mouse growth plate homeostasis through IHH signaling.J Bone Miner Res. 2024 Oct 29;39(11):1644-1658. doi: 10.1093/jbmr/zjae144. J Bone Miner Res. 2024. PMID: 39236220 Free PMC article.
-
mir-374-5p, mir-379-5p, and mir-503-5p Regulate Proliferation and Hypertrophic Differentiation of Growth Plate Chondrocytes in Male Rats.Endocrinology. 2018 Mar 1;159(3):1469-1478. doi: 10.1210/en.2017-00780. Endocrinology. 2018. PMID: 29390136 Free PMC article.
-
ALK2 functions as a BMP type I receptor and induces Indian hedgehog in chondrocytes during skeletal development.J Bone Miner Res. 2003 Sep;18(9):1593-604. doi: 10.1359/jbmr.2003.18.9.1593. J Bone Miner Res. 2003. PMID: 12968668
-
Regulatory mechanisms for the development of growth plate cartilage.Cell Mol Life Sci. 2013 Nov;70(22):4213-21. doi: 10.1007/s00018-013-1346-9. Epub 2013 May 4. Cell Mol Life Sci. 2013. PMID: 23640571 Free PMC article. Review.
-
Non-Coding RNAs in Cartilage Development: An Updated Review.Int J Mol Sci. 2019 Sep 11;20(18):4475. doi: 10.3390/ijms20184475. Int J Mol Sci. 2019. PMID: 31514268 Free PMC article. Review.
Cited by
-
miR-708-5p Attenuates Osteoarthritis Progression via Multi-Target Modulation of the NOX4/NF-κB Axis and Cartilage Homeostasis.Cartilage. 2025 Jul 30:19476035251361679. doi: 10.1177/19476035251361679. Online ahead of print. Cartilage. 2025. PMID: 40734512 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

