Ascorbic Acid 2-Phosphate-Releasing Supercritical Carbon Dioxide-Foamed Poly(L-Lactide-Co-epsilon-Caprolactone) Scaffolds Support Urothelial Cell Growth and Enhance Human Adipose-Derived Stromal Cell Proliferation and Collagen Production
- PMID: 40226413
- PMCID: PMC11919108
- DOI: 10.1155/2023/6404468
Ascorbic Acid 2-Phosphate-Releasing Supercritical Carbon Dioxide-Foamed Poly(L-Lactide-Co-epsilon-Caprolactone) Scaffolds Support Urothelial Cell Growth and Enhance Human Adipose-Derived Stromal Cell Proliferation and Collagen Production
Abstract
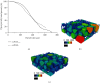
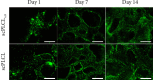
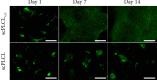
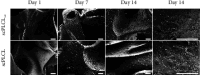
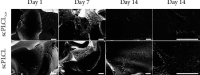
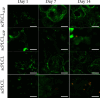
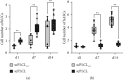
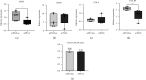
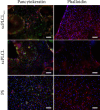
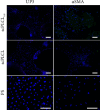
Tissue engineering can provide a novel approach for the reconstruction of large urethral defects, which currently lacks optimal repair methods. Cell-seeded scaffolds aim to prevent urethral stricture and scarring, as effective urothelium and stromal tissue regeneration is important in urethral repair. In this study, the aim was to evaluate the effect of the novel porous ascorbic acid 2-phosphate (A2P)-releasing supercritical carbon dioxide-foamed poly(L-lactide-co-ε-caprolactone) (PLCL) scaffolds (scPLCLA2P) on the viability, proliferation, phenotype maintenance, and collagen production of human urothelial cell (hUC) and human adipose-derived stromal cell (hASC) mono- and cocultures. The scPLCLA2P scaffold supported hUC growth and phenotype both in monoculture and in coculture. In monocultures, the proliferation and collagen production of hASCs were significantly increased on the scPLCLA2P compared to scPLCL scaffolds without A2P, on which the hASCs formed nonproliferating cell clusters. Our findings suggest the A2P-releasing scPLCLA2P to be a promising material for urethral tissue engineering.
Copyright © 2023 Alma Kurki et al.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures



Similar articles
-
Ascorbic Acid 2-Phosphate Releasing Supercritically Foamed Porous Poly-L-Lactide-Co-ε-Caprolactone Scaffold Enhances the Collagen Production of Human Vaginal Stromal Cells: A New Approach for Vaginal Tissue Engineering.Tissue Eng Regen Med. 2024 Jan;21(1):81-96. doi: 10.1007/s13770-023-00603-3. Epub 2023 Oct 31. Tissue Eng Regen Med. 2024. PMID: 37907765 Free PMC article.
-
Human Adipose Stem Cells Differentiated on Braided Polylactide Scaffolds Is a Potential Approach for Tendon Tissue Engineering.Tissue Eng Part A. 2016 Mar;22(5-6):513-23. doi: 10.1089/ten.tea.2015.0276. Epub 2016 Feb 26. Tissue Eng Part A. 2016. PMID: 26919401
-
Promoting cell proliferation and collagen production with ascorbic acid 2-phosphate-releasing poly(l-lactide-co-ε-caprolactone) membranes for treating pelvic organ prolapse.Regen Biomater. 2024 May 24;11:rbae060. doi: 10.1093/rb/rbae060. eCollection 2024. Regen Biomater. 2024. PMID: 38903561 Free PMC article.
-
Electrospun silk fibroin/poly(lactide-co-ε-caprolactone) nanofibrous scaffolds for bone regeneration.Int J Nanomedicine. 2016 Apr 11;11:1483-500. doi: 10.2147/IJN.S97445. eCollection 2016. Int J Nanomedicine. 2016. PMID: 27114708 Free PMC article.
-
Characterisation and in vitro and in vivo evaluation of supercritical-CO2-foamed β-TCP/PLCL composites for bone applications.Eur Cell Mater. 2019 Aug 5;38:35-50. doi: 10.22203/eCM.v038a04. Eur Cell Mater. 2019. PMID: 31381126
References
-
- Lazzeri M., Sansalone S., Guazzoni G., Barbagli G. Incidence, causes, and complications of urethral stricture disease. European Urology Supplements . 2016;15(1):2–6. doi: 10.1016/j.eursup.2015.10.002. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

