Optogenetic Activation of Intrinsic Cardiac Autonomic Neurons in Excised Perfused Mouse Hearts
- PMID: 40228035
- PMCID: PMC12276981
- DOI: 10.3791/67364
Optogenetic Activation of Intrinsic Cardiac Autonomic Neurons in Excised Perfused Mouse Hearts
Abstract
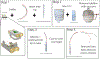
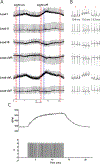
A balance of cholinergic and catecholaminergic activation is necessary to maintain heart health. Interrogating the interaction between these pathways can be done using optogenetics through selective expression of channelrhodopsin-2 (ChR2) in cardiac autonomic neurons. Such cardiac applications of optogenetics allow for the study of the intrinsic release of neurotransmitters in a spatiotemporal manner. This method illustrates an ex vivo approach for specific optogenetic stimulation of cardiac neurons in perfused mouse hearts. Transgenic mice were bred to express ChR2 in either choline acetyltransferase (ChAT) or tyrosine hydroxylase (TH) neurons throughout the body. A micro-LED (465 nm) encased in a silicone elastomer was prepared for stimulating the neurons of the right atrium that innervate the sinoatrial node. The micro-LED was connected to a function generator set to pulse waves at 10 Hz with a 30 ms pulse width. Hearts with confirmed expression were excised and retrogradely perfused on a Langendorff system circulating Krebs-Henseleit solution. Electrocardiogram (ECG), temperature, and coronary flow rate were recorded using the LabChart software. Once the heart stabilized, the micro-LED was placed on the right atrium and tested for optimal heart rate response. An application of this approach combines the intrinsic release of cholinergic neurotransmitter (acetylcholine) during optogenetic activation of a ChAT-ChR2 mouse heart simultaneously with increasing exogenous catecholaminergic neurotransmitter (norepinephrine) added to the perfusate. The resulting changes in heart rate during the simultaneous cholinergic and catecholaminergic activation are presented. This method describes a valuable experimental approach for investigating the kinetics of sudden intrinsic autonomic neuron activation in perfused hearts and the interactions between cardiac cholinergic and catecholaminergic activity.
Conflict of interest statement
Disclosures
No conflicts of interest to declare.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
