The Role of the Sirt1/Foxo3a Pathway in Mitigating Myocardial Ischemia-Reperfusion Injury by Dexmedetomidine
- PMID: 40230274
- PMCID: PMC11997638
- DOI: 10.1111/cbdd.70100
The Role of the Sirt1/Foxo3a Pathway in Mitigating Myocardial Ischemia-Reperfusion Injury by Dexmedetomidine
Abstract
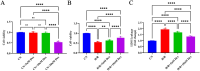
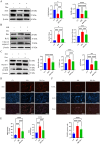
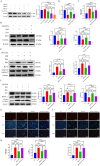
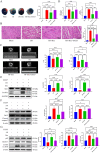
Myocardial ischemia-reperfusion injury (MIRI) significantly affects the prognosis of cardiac surgery patients. The anesthetic dexmedetomidine (Dex) has shown protective effects against ischemia-reperfusion injury in cardiomyocytes; however, its exact mechanism remains unclear. In this study, hypoxia/reoxygenation (H/R) and ischemia/reperfusion (I/R) models were used to investigate the effects of Dex on H9c2 cells and MIRI in mice. The roles of the Sirtuin 1/Forkhead box O3a (Sirt1/FoxO3a) pathway in the protective effects of Dex were explored using the Sirt1 inhibitor EX527 and FoxO3a gene silencing. Results showed that H/R significantly reduced H9c2 cell viability, increased Lactate Dehydrogenase (LDH) leakage, and elevated reactive oxygen species (ROS) production. Dex pretreatment reversed these effects. Additionally, Dex significantly reduced the expression of Bcl-2-associated X protein/B-cell lymphoma 2 (Bax/Bcl-2), cleaved caspase-3, Beclin-1, and microtubule-associated protein 1A/1B-light chain 3B (LC3B), inhibiting apoptosis and autophagy while increasing the expression of p62, Sirt1, and FoxO3a. The protective effects of Dex against H/R injury were abolished by EX527 or FoxO3a silencing. In the mouse MIRI model, Dex pretreatment decreased serum LDH and Creatine Kinase-MB (CK-MB) levels, reduced myocardial infarct size and cardiac injury, and significantly improved left ventricular ejection fraction (LVEF) and left ventricular fractional shortening (LVFS). These protective effects were markedly reversed by EX527. These findings indicate that Dex alleviates MIRI by restoring Sirt1 expression and activating FoxO3a.
Keywords: FoxO3a; Sirt1; apoptosis; autophagy; dexmedetomidine; myocardial ischemia–reperfusion injury.
© 2025 The Author(s). Chemical Biology & Drug Design published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Mir221- and Mir222-enriched adsc-exosomes mitigate PM exposure-exacerbated cardiac ischemia-reperfusion injury through the modulation of the BNIP3-MAP1LC3B-BBC3/PUMA pathway.Autophagy. 2025 Feb;21(2):374-393. doi: 10.1080/15548627.2024.2395799. Epub 2024 Sep 8. Autophagy. 2025. PMID: 39245438 Free PMC article.
-
Theaflavin-3,3'-digallate protects against myocardial ischemia/reperfusion injury and hypoxia/reoxygenation injury by activating the PI3K/Akt/mTOR pathway.J Mol Histol. 2025 Jun 27;56(4):207. doi: 10.1007/s10735-025-10453-z. J Mol Histol. 2025. PMID: 40576913
-
Dexmedetomidine attenuates ischemia and reperfusion-induced cardiomyocyte injury through p53 and forkhead box O3a (FOXO3a)/p53-upregulated modulator of apoptosis (PUMA) signaling signaling.Bioengineered. 2022 Jan;13(1):1377-1387. doi: 10.1080/21655979.2021.2017611. Bioengineered. 2022. Retraction in: Bioengineered. 2024 Dec;15(1):2299625. doi: 10.1080/21655979.2024.2299625. PMID: 34974801 Free PMC article. Retracted.
-
Adiponectin protects against myocardial ischemia-reperfusion injury: a systematic review and meta-analysis of preclinical animal studies.Lipids Health Dis. 2024 Feb 17;23(1):51. doi: 10.1186/s12944-024-02028-w. Lipids Health Dis. 2024. PMID: 38368320 Free PMC article.
-
A systematic review of p53 regulation of oxidative stress in skeletal muscle.Redox Rep. 2018 Dec;23(1):100-117. doi: 10.1080/13510002.2017.1416773. Epub 2018 Jan 3. Redox Rep. 2018. PMID: 29298131 Free PMC article.
Cited by
-
FoxO3 Activation Alleviates Doxorubicin-Induced Cardiomyopathy by Enhancing Autophagic Flux and Suppressing mTOR/ROS Signalling.J Cell Mol Med. 2025 Aug;29(15):e70775. doi: 10.1111/jcmm.70775. J Cell Mol Med. 2025. PMID: 40785055 Free PMC article.
References
-
- Chen, M. , Li X., and Mu G.. 2022. “Myocardial Protective and Anti‐Inflammatory Effects of Dexmedetomidine in Patients Undergoing Cardiovascular Surgery With Cardiopulmonary Bypass: A Systematic Review and Meta‐Analysis.” Journal of Anesthesia 36: 5–16. 10.1007/s00540-021-02982-0. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

