Cilostazole versus clopidogrel in acute large-vessel moderate and moderate-to-severe ischemic stroke: a randomized controlled trial
- PMID: 40232632
- PMCID: PMC12267345
- DOI: 10.1007/s10072-025-08107-9
Cilostazole versus clopidogrel in acute large-vessel moderate and moderate-to-severe ischemic stroke: a randomized controlled trial
Abstract
Background: More than one-third of all ischemic strokes are induced by large vessel occlusion (LVO). All the wide-scale trials that assessed the impacts of cilostazol versus clopidogrel in stroke management have been conducted in Asia and involved patients with minor stroke or TIA. Our trial is the first-ever study to evaluate cilostazol versus clopidogrel in acute LVO with moderate to severe ischemic stroke in North Africa.
Objectives: We assessed the efficacy and safety of cilostazol versus clopidogrel in first-ever LVO moderate and moderate to severe ischemic stroke patients.
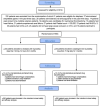
Methods: 580 moderate and moderate-to-severe LVO ischemic stroke participants were randomly enrolled to receive loading and maintenance doses of cilostazol or clopidogrel.
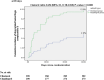
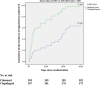
Results: 580 patients were included in the intention-to-treat analysis. 29 (10.0%) participants in the cilostazol arm and 43 (14.8%) participants in the clopidogrel arm experienced a new stroke (HR 0.37; 95% CI, 0.29-0.73; P-value = 0.03). Eight participants (2.8%) in the cilostazol arm and 17 patients (5.9%) in the clopidogrel arm had drug-related hemorrhagic complications (HR 0.29; 95% CI, 0.18-0.63; P-value = 0.008).
Conclusion: Patients who experienced acute LVO moderate and moderate-to-severe ischemic stroke and received loading and maintenance doses of cilostazol within the first 24 h after stroke onset had better clinical outcomes based on recurrent stroke rates and better safety outcomes regarding hemorrhagic transformation of brain infarction and drug-induced peripheral hemorrhagic side effects compared to those who received loading and maintenance doses of clopidogrel. There were no significant differences between the two groups regarding death due to vascular events and unfavorable mRS after three months of stroke onset.
Registration: Retrospectively registered on ClinicalTrials.gov, NCT06242145, 27-01-2024.
Keywords: Cilostazol; Clopidogrel; Egypt; Large-vessel stroke; Moderate ischemic stroke; Moderate-to-severe stroke.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Guarantor: Mohamed G. Zeinhom is the guarantor of the study. Ethics approval: Our study had the approval of the ethical committee of Kafr El-sheikh University, and the ethical reference number is (KFSIRB200-145). Consent to participate: Written informed consent was obtained from all subjects before the study. Consent for publication: Not applicable. Conflict of interest: Mohamed G. Zeinhom, Mohamed Fouad elsayed Khalil, Mohamed Ismaiel, Tarek youssif omar, Hossam Mohamed Refat, Ahmed ahmed Mohamed kamal ebied, Noha Abdelwahed, Ahmed Zaki Omar Akl, Emad Labib Abdelhamid Mahmoud, Salah Ibrahim Ahmed, Asmaa Mohammed Hassan, Islam Fathallah Mohamed Kamel, Amir Ahmed Elsaeed Egila, Mohamed Abouelnaga, and Sherihan Rezk ahmed declare that they have no potential conflicts of interest that might be relevant to the contents of this manuscript.
Figures


References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

