A variant of the autophagic receptor NDP52 counteracts phospho-TAU accumulation and emerges as a protective factor for Alzheimer's disease
- PMID: 40234443
- PMCID: PMC12000434
- DOI: 10.1038/s41419-025-07611-2
A variant of the autophagic receptor NDP52 counteracts phospho-TAU accumulation and emerges as a protective factor for Alzheimer's disease
Abstract
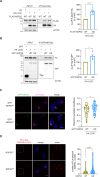
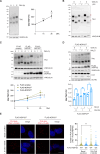
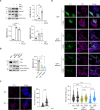
Selective elimination of early pathological TAU species may be a promising therapeutic strategy to reduce the accumulation of TAU, which contributes to neurodegeneration and is a hallmark of Alzheimer's disease (AD). Pathological hyper-phosphorylated TAU can be degraded through selective autophagy, and NDP52/CALCOCO2 is one of the autophagy receptors involved in this process. In 2021, we discovered a variant of NDP52, called NDP52GE (rs550510), that is more efficient at promoting autophagy. We here anticipate that this variant could be a powerful factor that could eliminate pathological forms of TAU better than its WT form (NDP52WT). Indeed, we provide evidence that in in vitro systems and in a Drosophila melanogaster model of TAU-induced AD, the NDP52GE variant is much more effective than the NDP52WT in reducing the accumulation of pathological forms of TAU through the autophagic process and rescues typical neurodegenerative phenotypes induced by hTAU toxicity. Mechanistically, we showed that NDP52WT and NDP52GE bind pTAU with comparable efficiency, but that NDP52GE binds the autophagic machinery (LC3C and LC3B) more efficiently than NDP52WT does, which could explain its greater efficiency in removing pTAU. Finally, by performing a genetic analysis of a cohort of 435 AD patients, we defined the NDP52GE variant as a protective factor for AD. Overall, our work highlights the variant NDP52GE as a resilience factor in AD that shows a robust effectiveness in driving pathological TAU degradation.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: The patients from IRCCS Santa Lucia Foundation Hospital of Rome were enrolled for a previous study approved by the Ethical Committee (CE/PROG.650 approved on 01/03/2018). The patients from the memory clinic of CRIC in Padua were collected according to the study protocol “Analisi delle Basi Biologiche della Malattia di Alzheimer eredofamiliare e delle forme sporadiche tardive” (3950/AO/2016) approved by the local ethic committee of the recruitment center on October 06th, 2016. All peripheral blood samples were collected after obtaining the written informed consent of the study participants, in accordance with the Helsinki Declaration.
Figures



References
-
- 2022 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2022;18:700–89. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

