High wall shear stress-dependent podosome formation in a novel murine model of intracranial aneurysm
- PMID: 40236952
- PMCID: PMC11999664
- DOI: 10.3389/fstro.2024.1494559
High wall shear stress-dependent podosome formation in a novel murine model of intracranial aneurysm
Abstract
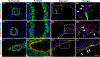
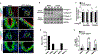
High wall shear stress (HWSS) contributes to intracranial aneurysm (IA) development. However, the underlying molecular mechanisms remain unclear, in part due to the lack of robust animal models that develop IAs in a HWSS-dependent manner. The current study established a new experimental IA model in mice that was utilized to determine HWSS-triggered downstream mechanisms. By a strategic combination of HWSS and low dose elastase, IAs were induced with a high penetrance in hypertensive mice. In contrast, no IAs were observed in control groups where HWSS was absent, suggesting that our new IA model is HWSS-dependent. IA outcomes were assessed by neuroscores that correlate with IA rupture events. Pathological analyses confirmed these experimental IAs resemble those found in humans. Interestingly, HWSS alone promotes the turnover of collagen IV, a major basement membrane component underneath the endothelium, and the formation of endothelial podosomes, subcellular organelles that are known to degrade extracellular matrix proteins. These induced podosomes are functional as they degrade collagen-based substrates locally in the endothelium. These data suggest that this new murine model develops IAs in a HWSS-dependent manner and highlights the contribution of endothelial cells to the early phase of IA. With this model, podosome formation and function was identified as a novel endothelial phenotype triggered by HWSS, which provides new insight into IA pathogenesis.
Keywords: cerebrovascular integrity; endothelial dysfunction; high wall shear stress; intracranial aneurysm; murine model; podosomes.
Conflict of interest statement
Conflict of interest The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

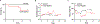


Similar articles
-
Flow-induced, inflammation-mediated arterial wall remodeling in the formation and progression of intracranial aneurysms.Neurosurg Focus. 2019 Jul 1;47(1):E21. doi: 10.3171/2019.5.FOCUS19234. Neurosurg Focus. 2019. PMID: 31261126 Free PMC article. Review.
-
Role of hemodynamics in initiation/growth of intracranial aneurysms.Eur J Clin Invest. 2018 Sep;48(9):e12992. doi: 10.1111/eci.12992. Epub 2018 Jul 20. Eur J Clin Invest. 2018. PMID: 29962043 Review.
-
Two Diverse Hemodynamic Forces, a Mechanical Stretch and a High Wall Shear Stress, Determine Intracranial Aneurysm Formation.Transl Stroke Res. 2020 Feb;11(1):80-92. doi: 10.1007/s12975-019-0690-y. Epub 2019 Feb 8. Transl Stroke Res. 2020. PMID: 30737656
-
Neutrophils: Novel Contributors to Estrogen-Dependent Intracranial Aneurysm Rupture Via Neutrophil Extracellular Traps.J Am Heart Assoc. 2023 Nov 7;12(21):e029917. doi: 10.1161/JAHA.123.029917. Epub 2023 Oct 27. J Am Heart Assoc. 2023. PMID: 37889179 Free PMC article.
-
Effect of Aneurysm and Patient Characteristics on Intracranial Aneurysm Wall Thickness.Front Cardiovasc Med. 2021 Dec 8;8:775307. doi: 10.3389/fcvm.2021.775307. eCollection 2021. Front Cardiovasc Med. 2021. PMID: 34957259 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
